Plasma Surface Modification: A Practical Guide to Activation, Functionalization & Wettability Control
By NineScrolls Engineering · 2026-04-26 · 24 min read · Plasma Processing
Target Readers: Surface scientists, polymer and biomaterials researchers, microfluidics and MEMS engineers, adhesion and coating specialists, and process engineers in medical device, automotive, packaging, and electronics industries who need to engineer surface chemistry rather than simply remove contamination. Newcomers will find the principles and gas-selection matrix grounding; experienced engineers can skip to the case data, hydrophobic-recovery countermeasures, and the cleaning-vs-modification comparison.
TL;DR
- Plasma surface modification is not plasma cleaning. Cleaning removes organic and particulate contamination; modification deliberately changes the surface's chemistry and energy state by introducing functional groups, breaking and crosslinking polymer chains, or grafting new species — the substrate is the target, not the contaminant.
- Three primary effects dominate: activation (raising surface energy from ~30 to >70 mN/m via –OH/–COOH/–NH2 generation), functionalization (selective grafting of specific chemistries — amine for biomolecule coupling, fluorocarbon for super-hydrophobicity), and crosslinking / CASING (Crosslinking by Activated Species of Inert Gases, which hardens and densifies the top nanometres).
- The working gas selects the chemistry: O2 drives oxidation and hydrophilicity; N2/NH3 introduce amine groups for bio applications; Ar creates physical activation and CASING; CF4/C4F8 deliver hydrophobic fluorocarbon termination; H2 reduces native oxides and hydrogen-terminates semiconductors.
- Real-world impact is large and quantifiable: PDMS-to-glass O2 plasma bonding routinely exceeds 300 kPa interfacial strength; PP automotive substrates jump from ASTM D3359 1B to 5B paint adhesion; Ti implants show 2–3× higher osteoblast attachment after combined O2/N2 activation.
- Hydrophobic recovery is the dominant pitfall. Untreated polymer surfaces drift back toward their bulk state within hours to days as low-molecular-weight chains migrate outward. Counter-strategies — immediate-use windows (≤30 min), refrigerated dry storage, PECVD SiOx capping, and reactive monomer post-grafting — are the difference between a lab demo and a manufacturable process.
- The same low-pressure RF plasma chamber used for cleaning can perform modification — what changes is the gas, the time, the power, and the post-process handling. Knowing the boundary lets one tool serve two distinct workflows.
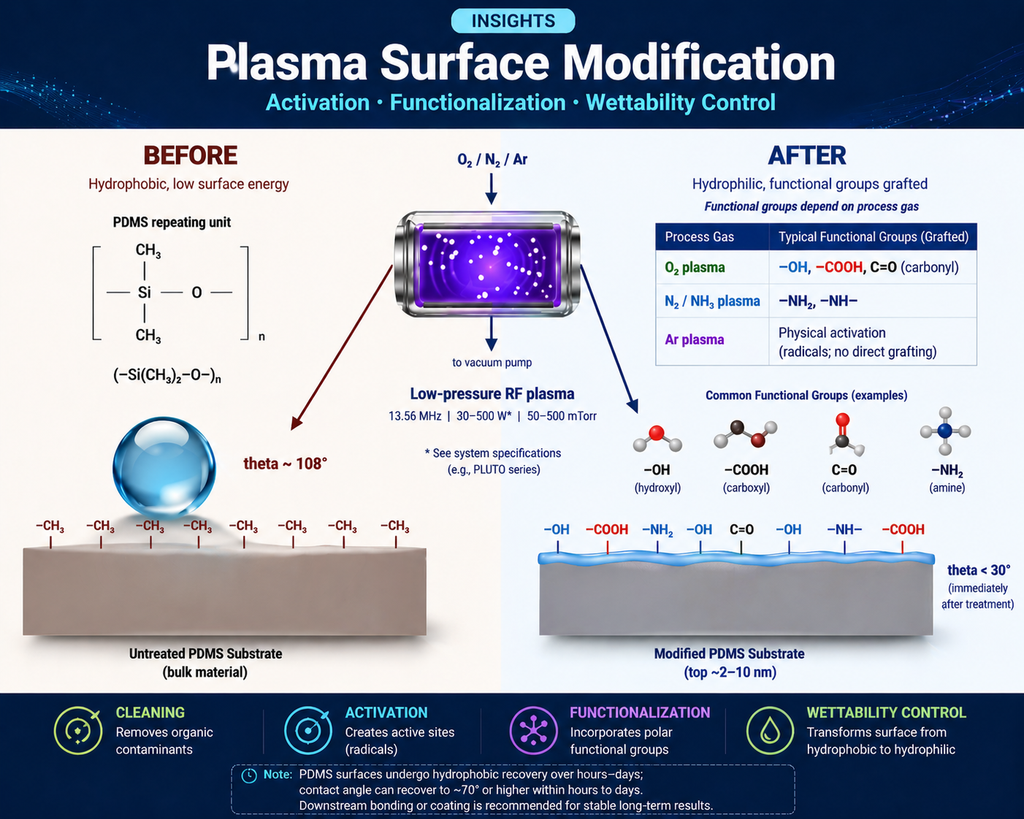
Figure 1: Plasma surface modification at a glance. Left: an untreated polymer surface with a contact angle >100°. Centre: low-pressure RF plasma reactor introducing reactive species. Right: the modified surface, now hydrophilic and decorated with –OH, –COOH, and –NH2 groups available for downstream bonding, coating, or biological functionalization.
1) What Plasma Surface Modification Is — and What It Is Not
Plasma is a partially ionized gas containing electrons, ions, radicals, excited neutrals, and UV photons. When this energetic mixture impinges on a solid surface, it can do four broadly distinguishable things: (1) physically sputter material away, (2) chemically etch material away, (3) deposit new material from precursor fragments, and (4) chemically modify the existing surface without net mass change. Plasma surface modification is the fourth category. The surface stays — but its top few nanometres are transformed.
This distinction matters because the equipment, the gas, and the runtime overlap heavily with plasma cleaning, plasma etching, and PECVD, and engineers routinely confuse them. A 30-second O2 plasma can be a cleaning step (if a contaminant film is present), an activation step (if the goal is to raise surface energy on a freshly molded part), or both. Calling it the right thing — and pairing it with the right downstream process — is what separates a robust workflow from one that drifts.
1.1 The Boundary with Plasma Cleaning
In plasma cleaning, the substrate is incidental and the contaminant is the target. The success criterion is contaminant removal, typically validated by water contact angle (a clean polymer wets) or by XPS (carbon contamination peak shrinks). In plasma surface modification, the contaminant is incidental — usually already removed by an upstream solvent wash — and the substrate itself is being chemically transformed. The success criterion is the appearance of new functional groups, a measurable change in surface energy, or a step-change in downstream behaviour (bonding strength, cell attachment, ink wetting).
Practically, the first minute of an O2 plasma on a typical polymer is doing both jobs at once: removing surface contamination and oxidizing the freshly exposed chains. After that, modification dominates. Knowing where the inflection sits for your specific material lets you pick the right time budget — too short and you have not built up enough functional groups; too long and you risk over-oxidation, surface roughening, or a brittle low-molecular-weight oxidized layer (LMWOL) that delaminates under load.
1.2 The Boundary with Etching and Deposition
Reactive ion etching uses the same plasma physics but selects gas chemistries (SF6, Cl2, fluorocarbons) and bias conditions that drive net material removal. PECVD adds vapor-phase precursors (SiH4, organosilicon monomers) that polymerize on the surface. Surface modification sits between them: enough plasma energy to break bonds and create reactive sites, but no precursor flux for film growth and no aggressive etchant for material removal. Ar, O2, N2, and small amounts of H2, NH3, or CF4 are the typical working gases, run at modest power and short to moderate times.
Key takeaway: If the surface composition before and after differs but the substrate dimension is unchanged within nanometres, you are doing modification — not cleaning, not etching, not deposition.
2) Three Primary Effects: Mechanism and Engineering Value
2.1 Activation — Raising Surface Energy
Activation is the most common reason engineers reach for a plasma. An inert polymer surface (PE, PP, PTFE, PDMS, polyolefin films) typically has a surface energy in the 18–35 mN/m range. Adhesives, paints, inks, and biological fluids all wet poorly under 50 mN/m. After 10–60 seconds of O2 or air plasma, polar groups (–OH, –COOH, C=O) appear in the top 2–10 nm and surface energy jumps to 50–75 mN/m. Contact angles fall from >90° to <20°. Bond strengths in downstream lap-shear or peel tests typically increase 3–10×.
The mechanism is straightforward radical chemistry: an O2 plasma generates atomic oxygen and OH radicals; these abstract hydrogen from the polymer (RH + O• → R• + •OH); the carbon-centred radical reacts with O2 to form a peroxy radical (R• + O2 → ROO•); the peroxy decomposes via β-scission, leaving terminal carbonyl, carboxyl, and hydroxyl groups. The net effect is a thin polar layer covalently bound to the substrate.
Engineering value, with numbers:
- Automotive PP bumpers before paint: 30 s air plasma raises adhesion from ASTM D3359 class 1B (large flake-off) to class 5B (no removal). This is the de-facto pretreatment in every major OEM paint line.
- Composite repair bonding: O2 plasma on CFRP raises lap-shear strength on epoxy adhesives by 60–120% versus solvent wipe alone.
- Lithium-ion separator wetting: 20–40 s O2/Ar plasma on PE/PP separator membranes drops electrolyte contact angle from ~50° to <10°, accelerating wet-out and reducing formation cycle time.
Key takeaway: Activation is the workhorse use case — short times, oxygen-bearing gas, dramatic and easily measured improvement in any downstream wetting or bonding step.
2.2 Functionalization — Grafting Specific Chemistries
Where activation aims for "more polar groups, any kind," functionalization aims for "specific groups, in known density." The gas chemistry is selected to leave a particular terminal functionality dominant. The difference matters when the next process step is a chemical reaction — covalent coupling of an antibody, electroless plating, ATRP polymer brush growth, or a click-chemistry handle.
- Amine functionalization (–NH2) via NH3, N2/H2, or N2 plasma is the standard route for biomolecule immobilization. PDMS microfluidic channels are routinely amine-functionalized then reacted with glutaraldehyde to anchor antibodies for biosensors. Typical surface density: 2–6 NH2 groups per nm2.
- Fluorocarbon functionalization (–CFx) via CF4, C4F8, or SF6/Ar plasma yields water contact angles of 130–160° on textured surfaces — used for textile water repellency, anti-stiction layers in MEMS release, and low-friction medical coatings.
- Carboxyl functionalization (–COOH) via O2 plasma followed by mild humidity exposure provides EDC/NHS-coupling sites, used for peptide conjugation onto polystyrene microspheres and ELISA plates.
- Hydrogen termination via H2 plasma on Si removes native oxide and leaves a hydrophobic Si–H surface — critical preparation step before low-temperature epitaxy or ALD nucleation control.
Key takeaway: If your downstream process is a specific chemical reaction, functionalization gives you the right handle at the right density. The gas determines the chemistry; power and time determine the density.
2.3 Crosslinking and CASING — Hardening the Top Surface
CASING — Crosslinking by Activated Species of Inert Gases — was first described in the 1960s and remains the cleanest way to densify a polymer surface without changing its bulk chemistry. An Ar (or He) plasma generates VUV photons (104–106 nm) and energetic metastables but no reactive radicals from the gas itself. These break C–H bonds in the polymer, generating carbon-centred radicals that recombine with neighbouring radicals to form new C–C crosslinks. The result is a 2–10 nm densified, crosslinked skin with higher modulus, lower oxygen permeability, and improved abrasion and chemical resistance.
- UHMWPE for joint implants: Ar plasma plus thermal annealing crosslinks the bearing surface, reducing wear debris generation in hip and knee prostheses by 40–80% in pin-on-disk testing.
- Polyimide barrier films: Ar/He plasma reduces oxygen transmission rate by 30–60%, useful in flexible OLED encapsulation stacks.
- Photoresist hardening: a brief Ar or He plasma "descum and crosslink" before deep etching improves resist erosion resistance by 15–30%.
CASING is also the mechanism behind the embrittled "skin" that develops on PDMS after long Ar or O2 exposure — a useful effect for stamps and replica molds, but a failure mode for stretchable electronics where the brittle skin cracks under strain.
Key takeaway: Inert-gas plasmas modify without grafting new chemistry. They harden, densify, and crosslink. Use them when you want mechanical or barrier improvement without changing surface energy.
3) Working Gas Selection — Reaction Mechanism Matrix
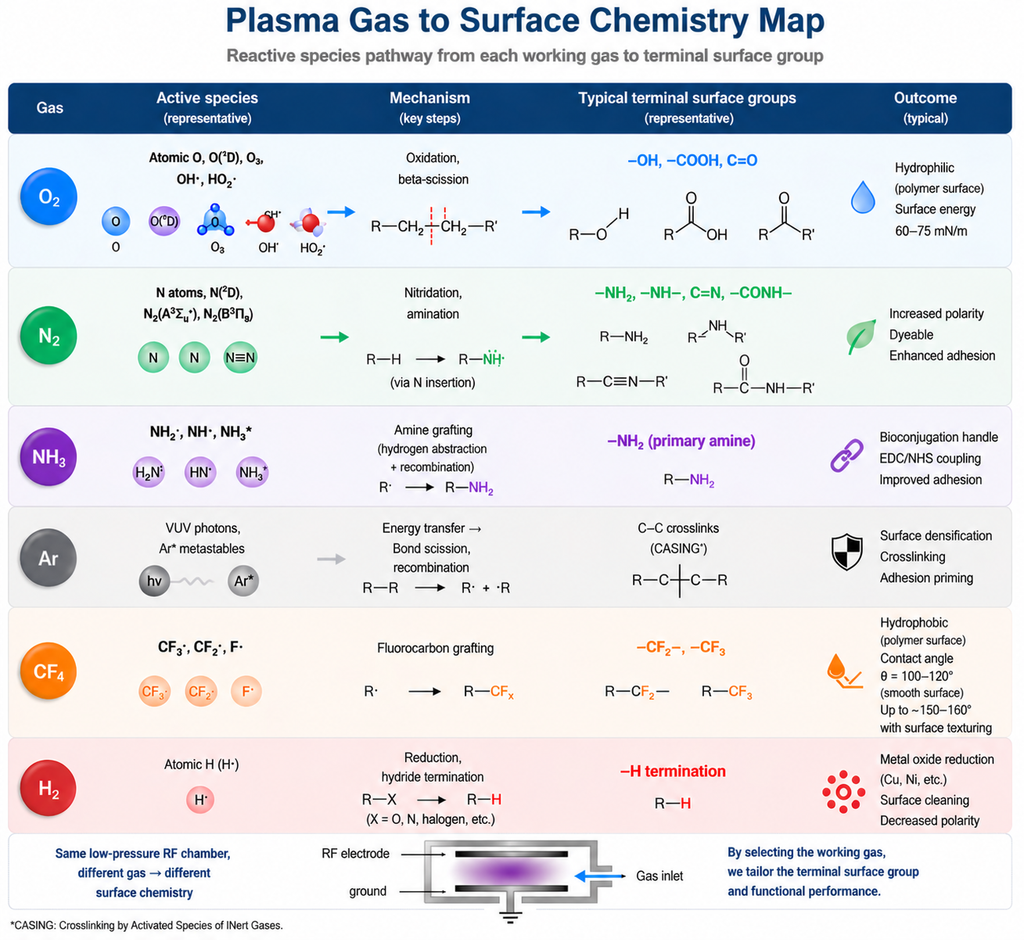
Figure 2: Plasma working gas → surface chemistry map. Each gas drives a distinct radical or ionic pathway, ending in a different terminal functional group on the substrate.
| Gas | Primary Reaction Path | Terminal Groups | Surface Effect | Typical Materials |
|---|---|---|---|---|
| O2 | RH + O• → R• + •OH; R• + O2 → ROO• → β-scission | –OH, –COOH, C=O | High polarity, contact angle <20°, surface energy 60–75 mN/m | PE, PP, PDMS, PET, PMMA |
| N2 | RH + N• → R–N• → R–N=N–R or R–C≡N | –C≡N, –N=N–, secondary amines | Moderate polarity, dyeability, some bio-affinity | PET, polyolefins, silk |
| NH3 | NH3 → •NH2 + •H; surface –C–NH2 attachment | –NH2 (primary amine, dominant) | Bioconjugation handle, EDC/NHS coupling, electroless plating | PDMS microfluidics, PET biosensors |
| Ar (or He) | VUV + ion-induced bond scission → C• → C–C recombination | No new chemistry — crosslinks (CASING) | Surface densification, hardening, barrier improvement | UHMWPE, polyimide, photoresist |
| CF4 / C4F8 | CFx• grafting onto C• sites; –CF2– / –CF3 termination | –CF2–, –CF3 | Hydrophobic / oleophobic; contact angle 110–160°; low friction | Textiles, MEMS anti-stiction, medical guidewires |
| H2 | H• + Si–O → Si–H + OH•; reduction of native oxide; metal de-oxidation | –H termination on Si; oxide-free metals | Hydride passivation, ALD nucleation prep, Cu surface activation | Si, Ge, Cu, GaAs, Pd |
Key takeaway: The gas is the chemistry. Power, pressure, and time are the dosage. Pick the gas first based on what terminal group your downstream step needs.
4) Quantitative Case Studies
4.1 PDMS-to-Glass O2 Plasma Bonding for Microfluidics
The most widely replicated plasma surface modification in academic labs. PDMS bulk surface energy is ~22 mN/m and contact angle ~108°. After a brief O2 plasma, surface –Si–OH groups form on both PDMS and glass; brought into contact within minutes, they condense to Si–O–Si covalent bonds across the interface.
Recipe (typical lab tool):
- Process: O2 plasma in a 13.56 MHz capacitively coupled cleaner
- Power: 30 W (low — high power over-oxidizes and weakens bonds)
- Pressure: 100 mTorr (~13 Pa)
- Flow: 20–50 sccm O2
- Time: 30 seconds on PDMS, 30 seconds on glass slide (in same run)
- Bond: bring surfaces into conformal contact within 1–5 min; light pressure; no further bake required
Measured outcomes:
- Contact angle on PDMS: 108° → <10° immediately post-plasma
- XPS O/C ratio on PDMS surface: ~0.5 → ~0.9 (silanol-enriched)
- Burst pressure of bonded channel: typically 250–400 kPa (3–6× untreated)
- T-peel strength: 4–8 N/cm depending on PDMS curing ratio
Failure modes: waiting more than ~30 minutes between plasma and contact dramatically weakens the bond (hydrophobic recovery rationale below); over-powered or over-long plasma generates a brittle silica-like skin that cracks under flexural load; plasma on under-cured PDMS creates a sticky surface that bonds inconsistently.
4.2 Polypropylene Bumper Pretreatment for Paint Adhesion
Untreated injection-molded PP has a surface energy near 30 mN/m and is essentially un-paintable with conventional waterborne basecoats. The standard line treatment is either flame treatment or atmospheric-pressure air plasma (an alternative low-pressure RF process is used for high-value parts).
Low-pressure RF process for engineering trials:
- Gas: O2 80%, Ar 20% (Ar improves uniformity on complex 3D shapes)
- Power: 200–400 W (scaled to chamber and load)
- Pressure: 200 mTorr
- Time: 30–60 seconds
Measured outcomes:
- Surface energy: 30 → 60–68 mN/m
- Cross-hatch adhesion (ASTM D3359): 1B → 5B
- Wet adhesion after 240 h humidity exposure: maintained at 4B–5B versus 0B for untreated
Process note: the activation has a useful window of about 24–72 hours under clean storage before paint application. Lines with longer queue times typically re-activate immediately before basecoat.
4.3 Titanium Implants — Osteoblast Attachment
Sandblasted, acid-etched (SLA) titanium implants are the orthopedic and dental industry baseline. A combined O2/N2 low-pressure plasma immediately before sterile packaging restores the surface to a fully hydrophilic state and introduces both –OH and –NH2/–NH groups, both of which protein-coat preferentially.
Recipe:
- Step 1: O2 plasma, 100 W, 200 mTorr, 60 s — strip carbon contamination, build silanol/hydroxyl
- Step 2: N2 plasma, 80 W, 250 mTorr, 30 s — introduce nitrogen functionalities
- Immediate vacuum-seal in moisture-impermeable foil
Measured outcomes:
- Static water contact angle: 70–90° (aged SLA) → <10° (fresh after plasma)
- Osteoblast (MC3T3-E1) attachment density at 4 h: 2.5–3× higher than aged SLA control
- Alkaline phosphatase activity at day 7: ~40% higher
4.4 Polyester Textile — CF4 Plasma for Water Repellency
Industrial alternative to fluorochemical wet finishes for water-repellent fabrics, with much lower chemical waste.
- Gas: CF4, optionally with H2 diluent (5–10%) to suppress etching
- Power: 150–300 W
- Pressure: 300 mTorr
- Time: 60–180 s
Static water contact angle on woven PET typically rises from 95° to 140–150°. AATCC 22 spray rating jumps from 0 to 90–100. The treatment is not as durable as a covalent C6 fluorochemical finish — repellency drops over 5–10 wash cycles — but eliminates wet chemistry and is a fit for short-life or single-use applications.
Key takeaway: Numbers from the literature and production lines line up. Activation gains are 3–10× in adhesion; fluorocarbon grafting takes contact angles past 140°; bio-functionalization can multiply cell attachment by 2–3×. The dosage windows are narrow but reproducible.
5) Hydrophobic Recovery — The Dominant Failure Mode
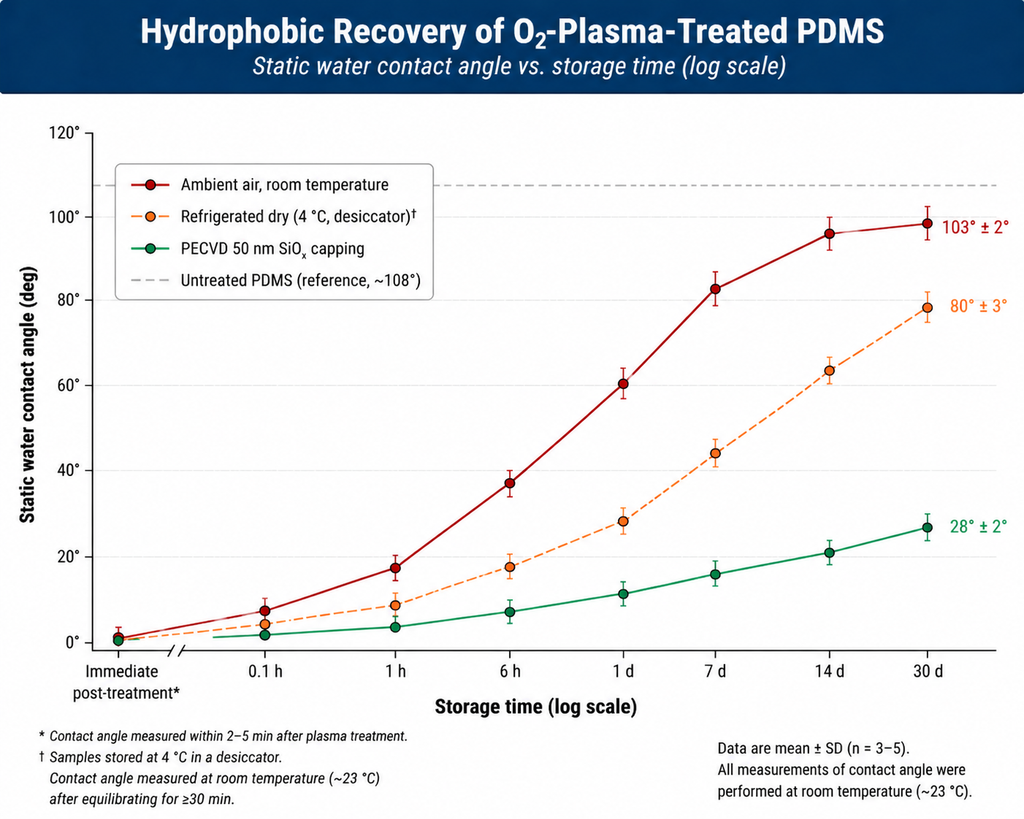
Figure 3: Hydrophobic recovery of O2-plasma-treated PDMS under three storage conditions over 30 days. Untreated reference at ~108° (dashed line); ambient storage drifts back to ~95° within a week; refrigerated dry storage roughly halves the recovery rate; a 50 nm PECVD SiOx capping layer locks the modified surface below 30° for the full month.
Surface activation is rarely permanent on polymers. Within hours to weeks, contact angles drift back upward and bonding strength falls. The mechanism is well understood: the bulk polymer contains low-molecular-weight chains and the freshly oxidized surface is in a high-energy, non-equilibrium state. Three pathways drive recovery:
- Chain rotation / reorientation. Polar groups on flexible chains rotate inward (away from the air interface) to lower interfacial free energy. This is the dominant mechanism in PDMS and other elastomers and operates on a timescale of minutes to hours.
- Migration of low-molecular-weight oligomers. Unreacted oligomers and short chains diffuse from the bulk to the surface, "burying" the modified layer. Most aggressive in soft, mobile-chain polymers like PDMS at room temperature.
- Diffusion of contaminants from air. Atmospheric hydrocarbons readsorb onto the high-energy surface, lowering it back. Negligible in clean rooms; significant in industrial environments.
5.1 Engineering Countermeasures (in order of cost)
| Strategy | Implementation | Effective Window | Cost / Complexity |
|---|---|---|---|
| Immediate-use window | Bond, coat, or coupling-react within 30 min of plasma | 30 min (PDMS); up to several hours (rigid polymers) | None — schedule discipline only |
| Refrigerated dry storage | Vacuum desiccator at 4 °C, dry N2 purge | 12–48 h (PDMS); 1–2 weeks (rigid polymers) | Low — desiccator + N2 line |
| Re-activation before use | Brief re-plasma (20–30 s O2) immediately before bonding/coating | Restores ~80–95% of original effect | Low — adds one short tool cycle |
| PECVD oxide capping | 10–50 nm SiOx via low-temp PECVD on top of activated surface | Indefinite (silica is stable); CA <30° for >30 days demonstrated | Medium — requires PECVD tool access |
| Reactive monomer grafting | Plasma-graft acrylic acid, allylamine, or PEG monomers post-activation | Months to years — covalently locked chemistry | High — additional gas/precursor handling, optimization |
Key takeaway: Recovery is not a defect — it is intrinsic to polymer thermodynamics. Plan around it: shorten the queue, store cold and dry, cap with PECVD, or graft a covalent layer.
6) Evaluation Methods — How to Prove It Worked
Surface modification is invisible to the naked eye and to most general-purpose lab tools. Choosing the right diagnostic for the question you are answering avoids both false positives (a clean-looking surface that is not actually functionalized) and false negatives (a properly modified surface that fails an inappropriate test).
| Method | What It Measures | Best Used For | Limitations |
|---|---|---|---|
| Static water contact angle | Apparent surface energy (Young's eq.) | Quick activation check; recovery monitoring | Insensitive to chemistry detail; affected by roughness |
| Owens-Wendt / dyne pen | Surface energy magnitude (mN/m) | Production line QC, paint/ink shops | Pen ink contaminates surface; ±5 mN/m precision |
| XPS (ESCA) | Elemental composition + bonding state, top 5–10 nm | Confirming functional group identity and density | Slow, expensive, UHV — not for inline use |
| ATR-FTIR | Functional group vibrations, ~1 µm depth | Detecting –OH, –COOH, C=O on polymers | Depth too deep for nm-scale changes; needs reference spectrum |
| SEM / AFM | Topography, roughness change | Detecting over-treatment etching, CASING skin cracking | No chemical info; sample-prep dependent |
| Lap-shear / T-peel / 90° peel | Practical bond strength | Adhesion-driven processes; production validation | Composite of surface + adhesive + cure; not pure surface metric |
| Dye / fluorescent labelling | Density of specific functional groups (–NH2, –COOH) | Bioconjugation site quantification | Indirect; assumes 1:1 binding stoichiometry |
| OES (in-situ) | Plasma species emission lines, real-time | Process monitoring, end-point detection | Measures plasma, not surface — proxy only |
Recommended baseline characterization stack for a new modification process:
- Static contact angle before/after — quick sanity check
- XPS for elemental and chemical-state confirmation — done once per process recipe
- The actual downstream test (peel, cell attachment, ink wetting) — the only one that matters for the application
- Contact angle aging study at 1 h / 24 h / 7 d / 30 d — quantify recovery for your specific material and storage conditions
Key takeaway: Contact angle is fast but coarse; XPS is definitive but slow. The downstream functional test is what decides whether the process is good enough. Always include a recovery study before scaling up.
7) Plasma Cleaning vs Plasma Surface Modification — Side-by-Side
Many engineers run both processes on the same tool with subtly different recipes. The comparison below makes the boundary explicit.
| Dimension | Plasma Cleaning | Plasma Surface Modification |
|---|---|---|
| Goal | Remove organic, particulate, or oxide contamination | Change surface chemistry / energy / functional groups |
| Target | Contaminant film (substrate is incidental) | Substrate top 2–10 nm (contaminant assumed gone) |
| Typical time | 10 s – 5 min | 30 s – 15 min |
| Typical power (200 mm chamber) | 50–300 W | 30–500 W (gas-dependent) |
| Typical pressure | 100–500 mTorr | 50–500 mTorr |
| Typical gases | O2, Ar, O2/Ar, occasionally H2 | O2, N2, NH3, Ar, CF4, H2, mixtures |
| Substrate temperature rise | <30 °C | <50 °C (often <30 °C) |
| Effect lifetime | Immediate use; surface re-contaminates within hours | Hours to days (polymers); months+ (with capping/grafting) |
| Validation method | Contact angle <10°, XPS C1s reduction | XPS new functional groups, FTIR peaks, downstream functional test |
| Equipment overlap | Same low-pressure RF plasma chamber serves both — typically PLUTO or HY series | |
Key takeaway: One tool, two workflows. The recipe and the validation differ; the hardware does not. Run a contamination check first; pick cleaning if removal is the goal, modification if chemistry change is the goal.
8) Process Window — Power, Time, and the Sweet Spot
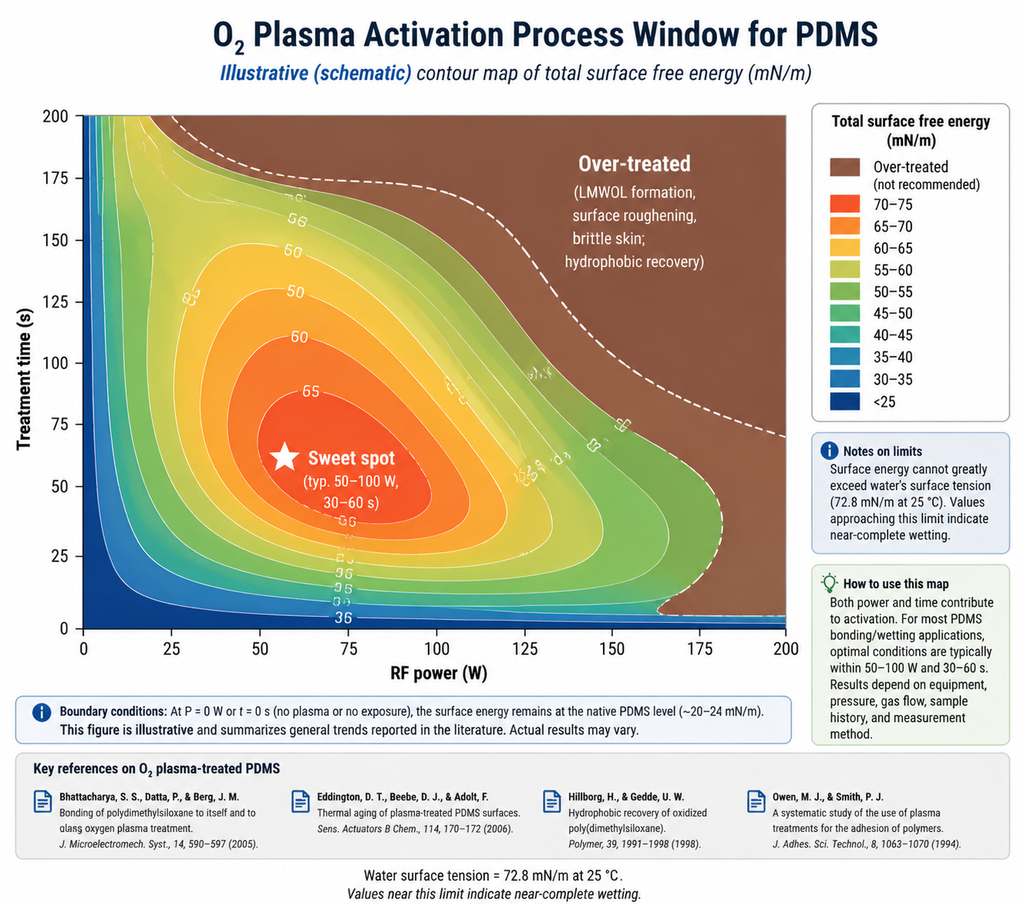
Figure 4: Process window for O2 plasma activation on PDMS. Surface energy contours (mN/m) as a function of RF power and treatment time. The shaded sweet spot at moderate power and short time gives high surface energy without LMWOL formation or surface roughening.
For most polymer activation processes, three regions exist on a power × time map:
- Under-treated (low power, short time): insufficient radical flux to fully oxidize the surface. Contact angle drops only partially; XPS shows weak –OH/–COOH peaks. Failure mode: inconsistent downstream bonding.
- Sweet spot (moderate power, short-to-moderate time): full activation, minimal substrate damage. This is the target operating window.
- Over-treated (high power, long time): radical flux exceeds the chain-scission threshold. A low-molecular-weight oxidized layer (LMWOL) builds up — full of polar groups but mechanically weak. Bond tests show cohesive failure within the LMWOL itself rather than at the interface. AFM reveals surface roughening; SEM may show micro-cracking after thermal cycling.
Practical defaults for new processes (200 mm chamber, 13.56 MHz CCP):
- Polymer activation: start 50 W, 100 mTorr O2, 30 s. Move power before time.
- Bio-amine functionalization: start 80 W, 200 mTorr NH3 or N2, 60 s.
- Hydrophobic CF4 grafting: start 200 W, 300 mTorr, 120 s.
- Inert-gas CASING: start 100 W, 100 mTorr Ar, 60–180 s.
For each new substrate, run a 3 × 3 DOE (low/mid/high power × short/mid/long time) and characterize with contact angle plus the downstream functional test. The optimum often differs from supplier-quoted recipes because chamber geometry, pumping speed, and electrode area all shift the effective dose.
Key takeaway: Move power before time when chasing higher activation; over-time creates LMWOL faster than over-power. Validate with the application test, not just contact angle.
9) Equipment Considerations
The same low-pressure RF plasma cleaner that serves as a contamination removal tool can perform the full range of surface modification processes covered in this guide, with two practical caveats: gas line plumbing and post-process handling.
9.1 Gas Plumbing
A baseline single-gas O2 system covers activation only. To unlock the full chemistry palette, the chamber needs:
- Multi-channel mass flow controllers — typically 3–5 channels (O2, N2, Ar, plus one or two specialty lines)
- Compatibility with corrosive gases (NH3, CF4) — appropriate seals, regulator materials, and exhaust scrubbing
- Mixing manifold for gas blends (O2/Ar, N2/H2) without back-diffusion
- Adequate pumping for fluorinated gases — turbomolecular or appropriately rated rotary vane
9.2 Power Source Choice
- 13.56 MHz RF (CCP) is the workhorse. Compatible with all materials including insulators. Moderate ion energy gives controllable activation without heavy substrate damage.
- 2.45 GHz microwave generates very high radical density with low ion energy — excellent for sensitive substrates (biological scaffolds, fragile electronics, ALD pre-treatment). Hardware is more specialized.
- DC plasma is incompatible with insulating substrates due to charge accumulation — limits its use in surface modification, where most targets are polymers or dielectrics.
- Atmospheric-pressure plasma jets are an alternative for inline production (textile, automotive paint lines) but offer less control over chemistry and dose; they sit outside the scope of this guide.
9.3 Substrate Handling
- Vented vacuum dry-storage cabinet adjacent to the tool — minimizes the air exposure window between unload and downstream use
- Inert-gas (N2) glove box for highest-stakes processes (bio-functionalization, fresh implant packaging)
- Automated load-locks for production tools to keep moisture and hydrocarbons out of the chamber base pressure
NineScrolls' PLUTO and HY series plasma cleaners support multi-gas configurations suitable for activation, functionalization, and CASING workflows; the same chambers are used for general cleaning, so a single tool can serve both workflows in a research lab — see the Plasma Cleaner Buying Guide for chamber sizing and gas-line option selection.
10) Frequently Asked Questions
What is the difference between plasma cleaning and plasma surface modification?
Plasma cleaning removes organic and particulate contamination from a surface — the substrate is incidental and the success criterion is contaminant removal. Plasma surface modification deliberately changes the substrate's own chemistry by introducing functional groups, crosslinking the top layer, or grafting new species. The same equipment runs both processes; what differs is the gas, the time, the power, and what is measured afterward.
How long does plasma surface activation last?
On polymers, activation is partially reversed within hours to days due to chain reorientation and migration of low-molecular-weight chains — this is hydrophobic recovery. PDMS recovers from <10° to ~95° within roughly a week at ambient. Refrigerated dry storage doubles to triples that lifetime. PECVD silica capping or covalent monomer grafting can lock the modification in place essentially indefinitely. On rigid polymers (PEEK, polyimide) recovery is far slower — weeks rather than hours.
Which gas should I use to activate a polymer surface for paint or adhesive bonding?
Start with O2, optionally with 10–20% Ar for uniformity on complex 3D parts. Oxygen plasma generates –OH, –COOH, and C=O groups that drive both polar interactions and covalent bonding with most adhesives. If your adhesive system is amine-cured (some polyurethanes and epoxies), an N2 or NH3 finishing step adds primary amine groups for stronger covalent attachment.
Can I do plasma surface modification on metals?
Yes, and it is increasingly common. H2 plasma reduces native oxides on Cu, GaAs, and similar surfaces — used as an in-situ pre-clean before low-temperature soldering, wire bonding, or ALD nucleation. O2/N2 plasma on Ti raises hydrophilicity and biocompatibility for medical implants. Ar plasma sputter-cleans noble metals (Au, Pt) prior to thin-film deposition.
Why does my PDMS bond fail when I delay between plasma and contact?
Hydrophobic recovery. The Si–OH groups generated by O2 plasma on PDMS are required to condense across the interface into Si–O–Si bonds when contacted with another silanol-bearing surface. Within minutes those silanol groups begin to be buried by chain reorientation and oligomer migration, and within an hour the bond strength is significantly degraded. Best practice: bond within 5 minutes of plasma; never longer than 30 minutes.
How do I know if my surface is over-treated?
Three signs: (1) contact angle that initially drops to a low value but rebounds upward within minutes — indicating a brittle LMWOL that washes or wipes off; (2) cohesive bond failure within the modified layer rather than at the interface, visible by SEM cross-section; (3) AFM showing roughness increase >5–10 nm. Drop power before time when troubleshooting; LMWOL forms faster from prolonged exposure than from higher power within reasonable bounds.
Is atmospheric-pressure plasma suitable for surface modification?
For activation in inline production environments — yes, and it is widely deployed in automotive, packaging, and textile lines. For controlled functionalization with specific chemistries (–NH2, –CFx) or for crosslinking-only CASING processes, low-pressure RF gives much better dose uniformity, gas chemistry control, and reproducibility. Atmospheric jets are best when throughput dominates; low-pressure systems are best when precision dominates.
Does plasma surface modification damage the bulk material?
The chemistry change is confined to the top 2–10 nm — far less than 1% of the thickness of any practical substrate. Bulk mechanical properties are unchanged. The only practical "damage" modes are surface roughening or micro-cracking under aggressive over-treatment (the LMWOL or CASING-skin failure described above), and these are avoidable with a properly bounded process window.
11) Related Reading
- What Is a Plasma Cleaner — Principles and Types — fundamentals of low-pressure RF plasma sources used for both cleaning and modification
- Plasma Cleaner Applications Guide — industry-by-industry survey of cleaning use cases that frequently overlap with the modification recipes covered here
- Plasma Cleaner Buying Guide — chamber sizing, gas-line options, and power source selection for multi-purpose tools
- PECVD Complete Guide — for silica capping layers that lock in plasma-activated surface chemistry
- ALD Comprehensive Guide — for functionalized surfaces serving as ALD nucleation templates
12) References
- Chu, P. K., Chen, J. Y., Wang, L. P. & Huang, N. "Plasma-surface modification of biomaterials." Materials Science and Engineering: R, 36(5–6), 143–206 (2002). doi:10.1016/S0927-796X(02)00004-9
- Hegemann, D., Brunner, H. & Oehr, C. "Plasma treatment of polymers for surface and adhesion improvement." Nuclear Instruments and Methods in Physics Research B, 208, 281–286 (2003). doi:10.1016/S0168-583X(03)00644-X
- Bhattacharya, S., Datta, A., Berg, J. M. & Gangopadhyay, S. "Studies on surface wettability of poly(dimethyl) siloxane (PDMS) and glass under oxygen-plasma treatment and correlation with bond strength." Journal of Microelectromechanical Systems, 14(3), 590–597 (2005). doi:10.1109/JMEMS.2005.844746
- Schwartz, J., Avaltroni, M. J., Danahy, M. P., Silverman, B. M., Hanson, E. L., Schwarzbauer, J. E., Midwood, K. S. & Gawalt, E. S. "Cell attachment and spreading on metal implant materials." Materials Science and Engineering: C, 23(3), 395–400 (2003).
- Owens, D. K. & Wendt, R. C. "Estimation of the surface free energy of polymers." Journal of Applied Polymer Science, 13(8), 1741–1747 (1969).
- Schonhorn, H. & Hansen, R. H. "Surface treatment of polymers for adhesive bonding." Journal of Applied Polymer Science, 11(8), 1461–1474 (1967). [Original CASING reference.]
- ASTM D3359-23: Standard Test Methods for Rating Adhesion by Tape Test. astm.org