Atomic Layer Deposition (ALD) – Principles, Process Windows, Materials, and Equipment Guide
By NineScrolls Engineering · 2026-03-27 · 18 min read · Materials Science
Target Readers: Semiconductor/MEMS process engineers, thin-film researchers, equipment engineers, PIs/lab managers, and R&D procurement teams evaluating ALD systems. Newcomers will find the fundamentals and glossary helpful; experienced engineers can jump to the precursor tables, process optimization, and equipment selection sections.
TL;DR Summary
Atomic Layer Deposition (ALD) is a vapor-phase thin-film technique that deposits material one atomic layer at a time through sequential, self-limiting surface reactions. This self-limiting nature delivers unmatched thickness control (<1 Å precision), exceptional conformality (>98% step coverage on high-aspect-ratio features), and sub-percent uniformity across wafers up to 300 mm. ALD is the enabling deposition technology for sub-5 nm gate dielectrics, 3D NAND passivation, MEMS encapsulation, and next-generation energy devices. This guide covers the reaction mechanism, compares thermal and plasma-enhanced ALD, maps the ALD temperature window, tabulates precursors and growth rates for >15 materials, benchmarks ALD against CVD/PVD/PECVD/MBE, and provides a practical framework for process optimization, troubleshooting, and equipment selection.
1) What is Atomic Layer Deposition?
Atomic Layer Deposition (ALD) is a chemical vapor deposition variant in which two or more gaseous precursors are introduced sequentially into a reaction chamber, separated by purge steps. Each precursor reacts with the substrate surface in a self-limiting manner: once every available surface site has reacted, no further deposition occurs regardless of additional precursor exposure. This self-limiting chemistry is the defining feature that distinguishes ALD from conventional CVD, where precursors react continuously in the gas phase or on the surface without an inherent stopping mechanism.
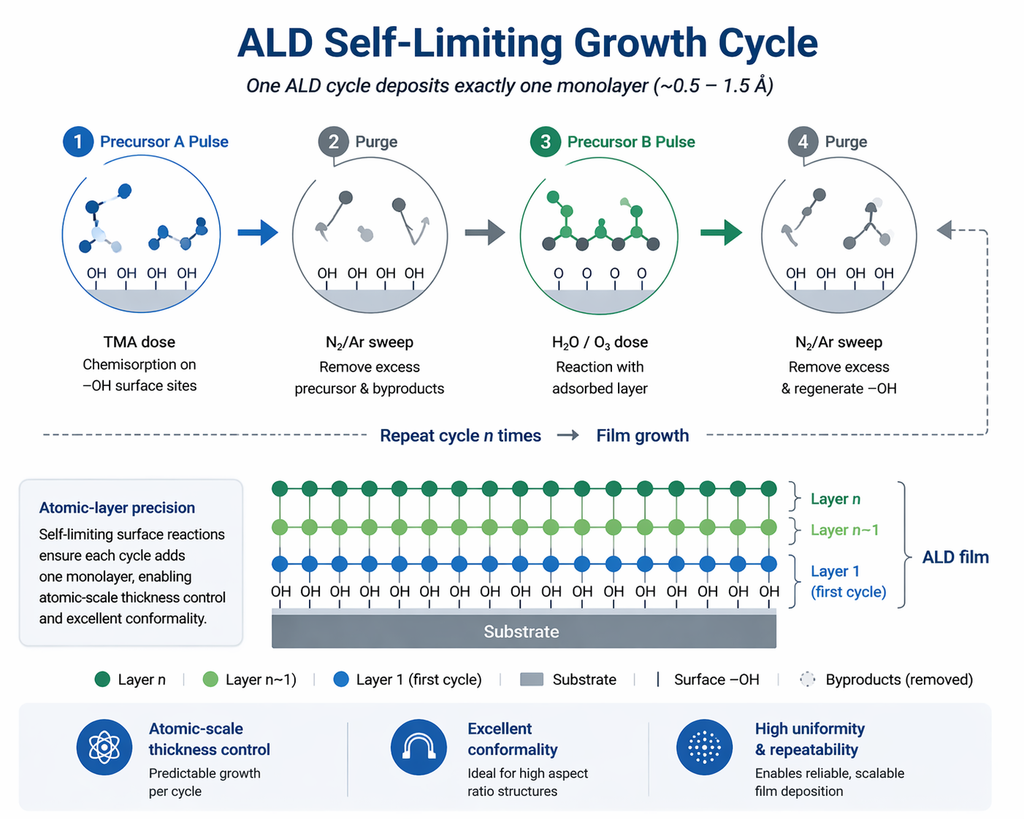
The Four-Step ALD Cycle
A single ALD cycle consists of four sequential steps, repeated hundreds or thousands of times to build the desired film thickness:
- Precursor A pulse: The first precursor (e.g., trimethylaluminum, TMA) is introduced into the chamber. It chemisorbs onto reactive surface sites (e.g., hydroxyl groups) through ligand-exchange or dissociative adsorption. Once all available sites are occupied, the reaction self-terminates.
- Purge 1: An inert gas (N₂ or Ar) sweeps away unreacted precursor and volatile by-products (e.g., CH₄ from TMA). Adequate purge time is critical to prevent CVD-like gas-phase reactions.
- Precursor B pulse (co-reactant): The second reactant (e.g., H₂O, O₃, O₂ plasma, or NH₃) reacts with the chemisorbed surface species, completing the formation of one sub-monolayer of the target material (e.g., Al₂O₃) and regenerating surface sites for the next cycle.
- Purge 2: A second inert gas purge removes excess co-reactant and by-products, resetting the surface for the next Precursor A pulse.
The net result is the deposition of a precisely controlled fraction of a monolayer per cycle, typically 0.5–2 Å depending on the material system and process temperature.
Why Self-Limiting Chemistry Matters
The self-limiting nature of ALD reactions provides several fundamental advantages that no other deposition technique can match simultaneously:
- Sub-angstrom thickness control: Film thickness is determined solely by the number of ALD cycles, not by precursor flux, exposure time, or substrate position. This digital control enables thickness precision better than ±1 Å.
- Exceptional conformality: Because each surface site reacts identically regardless of geometry, ALD coats the tops, bottoms, and sidewalls of high-aspect-ratio structures with equal thickness. Step coverage >98% is routinely achieved on aspect ratios of 100:1 or higher.
- Wafer-scale uniformity: Self-limiting chemistry inherently compensates for flux non-uniformities. Uniformity <1% (1σ) is standard across 200–300 mm wafers without complex gas-distribution engineering.
- Pinhole-free ultra-thin films: The layer-by-layer growth mode fills surface defects and grain boundaries, producing continuous, dense films even at thicknesses below 5 nm.
- Precise composition control: Ternary and quaternary alloys (e.g., HfAlOₓ, TiAlN) are synthesized by interleaving cycles of different binary ALD processes with arbitrary cycle ratios.
Brief History of ALD
ALD was independently developed in two contexts. In the 1960s and 1970s, Valentin Alekseevich Aleskovskii and Stanislav Koltsov at Leningrad Technological Institute explored “molecular layering” (ML) for depositing metal oxides. In parallel, Tuomo Suntola in Finland invented Atomic Layer Epitaxy (ALE) in 1974 for growing ZnS and Al₂O₃ thin films for electroluminescent flat-panel displays. Suntola’s work, patented in 1974 (Finnish patent 52,359), is widely credited as the origin of modern ALD. The technique remained a niche technology until the early 2000s, when Intel adopted ALD-grown HfO₂ as a high-κ gate dielectric at the 45 nm node—a breakthrough that made ALD an indispensable tool in semiconductor manufacturing.
2) Thermal ALD vs Plasma-Enhanced ALD (PEALD)
ALD processes fall into two broad categories based on how the co-reactant delivers its reactive species: thermal ALD and plasma-enhanced ALD (PEALD). Both preserve the self-limiting character of ALD, but they differ in reactivity, temperature requirements, and substrate compatibility.
Thermal ALD
In thermal ALD, the co-reactant (e.g., H₂O, O₃, H₂O₂, NH₃) reacts with the chemisorbed precursor species purely through thermally activated chemistry. The substrate temperature provides the activation energy for the surface reaction.
- Advantages: Gentle process with no plasma damage; excellent conformality even in ultra-high-aspect-ratio structures; simple reactor design; well-suited for sensitive substrates (polymers, biological, OLED).
- Limitations: Requires higher substrate temperatures for some material systems; limited reactivity for depositing metals and some nitrides; slower growth rates for certain processes; H₂O-based processes suffer from long purge times due to surface hydroxyl retention.
Plasma-Enhanced ALD (PEALD)
In PEALD, a plasma discharge generates highly reactive radical species (O*, N*, H*, NHₓ*) from the co-reactant gas. These radicals provide significantly more chemical energy than thermal molecules, enabling reactions that are thermodynamically or kinetically inaccessible in thermal ALD.
- Advantages: Lower deposition temperatures (enabling coating of temperature-sensitive substrates); access to a wider range of materials (especially metals like Pt, Pd, W, Ru and nitrides like TiN, TaN, SiNₓ, AlN, GaN); higher film density and fewer impurities at low temperatures; shorter cycle times due to more efficient surface reactions.
- Limitations: Potential plasma damage to sensitive substrates or interfaces; reduced conformality in very high-aspect-ratio features (radical recombination on sidewalls); more complex reactor design; line-of-sight component to radical flux can reduce step coverage in extreme geometries.
Remote Plasma vs Direct Plasma
PEALD systems use two plasma configurations:
- Direct plasma: The plasma is generated between electrodes in the deposition chamber, directly above the substrate. Higher radical flux, but also higher ion bombardment energy on the substrate surface.
- Remote plasma: The plasma is generated in a separate upstream source (typically inductively coupled), and only the neutral radical species travel downstream to the substrate. This eliminates energetic ion bombardment, reducing substrate damage while maintaining the reactivity benefits of plasma chemistry. Remote plasma is strongly preferred for gate dielectric deposition and other damage-sensitive applications.
Thermal ALD vs PEALD Comparison
| Parameter | Thermal ALD | PEALD |
|---|---|---|
| Co-reactant | H₂O, O₃, H₂O₂, NH₃ | O₂ plasma, N₂ plasma, H₂ plasma, NH₃ plasma |
| Typical temperature | 150–400 °C | 25–300 °C |
| Step coverage (100:1 AR) | >98% | 85–98% (remote plasma); 70–90% (direct plasma) |
| Film density | Good at optimal temperature | Higher at low temperatures |
| Carbon impurities | Low at optimal temperature; increases at low T | Very low even at low T |
| Substrate damage | None | Low (remote) to moderate (direct) |
| Metal deposition | Limited (few thermal metal ALD processes) | Excellent (Pt, Pd, Ru, W, TiN, TaN) |
| Best for | High-AR structures, sensitive substrates, oxides | Metals, nitrides, low-T deposition, high-density films |
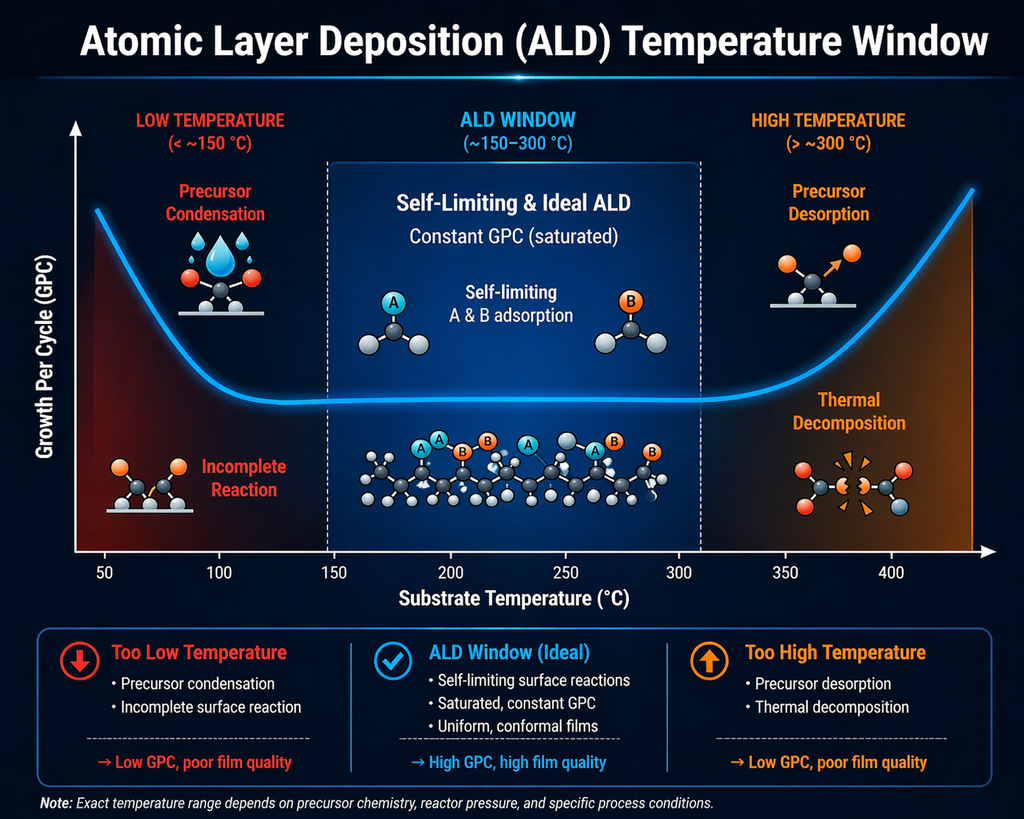
3) ALD Process Window & Temperature
Every ALD process has a characteristic ALD temperature window—a range of substrate temperatures within which the growth per cycle (GPC) is constant and the reactions are truly self-limiting. Understanding this window is essential for process development and optimization.
The ALD Temperature Window Explained
The ALD window is bounded by four failure modes:
- Below the window (low T, condensation regime): Precursor molecules physisorb and condense on the surface rather than chemisorbing. GPC increases uncontrollably, and films contain excess unreacted precursor and impurities.
- Below the window (low T, kinetic regime): Surface reactions are too slow to reach completion within the pulse time. GPC drops, and films are incomplete and non-uniform.
- Above the window (high T, decomposition regime): The precursor thermally decomposes before it can undergo the intended self-limiting surface reaction. GPC increases and the process reverts to CVD-like behavior with degraded uniformity and conformality.
- Above the window (high T, desorption regime): Chemisorbed precursor species or surface functional groups desorb before the co-reactant arrives. GPC drops, and films may be incomplete.
Within the ALD window, GPC is essentially constant and independent of pulse time (beyond the saturation dose), substrate position, and minor temperature fluctuations. This is the operating regime that delivers the hallmark benefits of ALD: digital thickness control, uniformity, and conformality.
Typical ALD Temperature Windows
| Material | Precursor | Co-reactant | ALD Window (°C) | GPC (Å/cycle) |
|---|---|---|---|---|
| Al₂O₃ | TMA | H₂O | 100–350 | 1.0–1.2 |
| HfO₂ | TDMAH / TEMAH / HfCl₄ | H₂O / O₃ | 200–350 | 0.9–1.1 |
| SiO₂ | BDEAS / 3DMAS / BTBAS | O₂ plasma | 50–300 (PEALD) | 0.8–1.4 |
| TiO₂ | TDMAT / TiCl₄ | H₂O / O₂ plasma | 100–300 | 0.4–0.6 |
| Ga₂O₃ | TMGa / TEGa | H₂O / O₂ plasma | 150–350 | 0.5–1.0 |
| ZnO | DEZ | H₂O | 100–250 | 1.5–2.0 |
| TiN | TDMAT / TiCl₄ | NH₃ / N₂/H₂ plasma | 200–400 | 0.4–0.6 |
| TaN | TBTDET / PDMAT | NH₃ / H₂ plasma | 200–350 | 0.3–0.5 |
| SiNₓ | BDEAS / DTDN2-H2 | N₂/Ar plasma | 200–400 (PEALD) | 0.3–1.0 |
| AlN | TMA | N₂/H₂ plasma / NH₃ | 200–400 | 0.8–1.1 |
| GaN | TMGa / TEGa | N₂/H₂ plasma / NH₃ plasma | 200–400 (PEALD) | 0.5–1.2 |
| Pt | MeCpPtMe₃ | O₂ plasma / O₃ | 200–350 | 0.4–0.5 |
| Pd | Pd(hfac)₂ | H₂ plasma / formalin | 150–300 | 0.2–0.5 |
| W | WF₆ / W(CO)₆ | Si₂H₆ / H₂ plasma | 200–350 | 0.5–1.5 |
| Ru | RuCp₂ / (EtCp)₂Ru | O₂ / O₂ plasma | 250–350 | 0.4–0.6 |
Extending Beyond the Thermal Window with PEALD
When a thermal ALD process requires temperatures incompatible with the device stack (e.g., polymer substrates that cannot tolerate >100 °C, or back-end-of-line interconnects limited to <200 °C), PEALD provides an alternative route. Plasma-generated radicals offer sufficient reactivity to drive surface reactions at temperatures 100–200 °C below the thermal ALD window, while maintaining growth rates within the 0.5–2 Å/cycle range. For example, high-quality SiO₂ can be deposited by PEALD at 50 °C using BDEAS + O₂ plasma, compared to >150 °C required for thermal approaches.
4) Common ALD Precursors & Materials
Precursor selection is one of the most critical decisions in ALD process development. An ideal ALD precursor must satisfy several stringent requirements:
- Sufficient vapor pressure: Must be volatile enough at practical source temperatures (20–200 °C) to deliver adequate dose to the reactor.
- Thermal stability: Must not decompose at the substrate temperature. Premature decomposition destroys the self-limiting mechanism.
- High reactivity with the surface: Must chemisorb aggressively and irreversibly onto the target surface sites.
- Clean ligand elimination: Reaction by-products must be volatile and non-contaminating. Carbon, chlorine, or nitrogen incorporation from incompletely removed ligands degrades film properties.
- No etching of the growing film: Some precursors or by-products (notably HCl from chloride-based precursors) can etch the deposited material, reducing GPC and causing non-uniformity.
Precursor Classification
| Precursor Class | Examples | Advantages | Limitations |
|---|---|---|---|
| Metal alkyls | TMA, DEZ, TMGa, TEGa | High reactivity, clean reactions, high vapor pressure | Pyrophoric (TMA, DEZ); limited to certain metals |
| Metal halides | TiCl₄, HfCl₄, WF₆, AlCl₃ | High thermal stability, well-studied | Corrosive by-products (HCl, HF); may etch films; particle generation |
| Metal amides | TDMAT, TDMAH, TEMAH, TBTDET | Moderate vapor pressure; no halide by-products | Lower thermal stability (decompose at ~350 °C); carbon incorporation risk |
| Metal cyclopentadienyls | RuCp₂, (EtCp)₂Ru, MeCpPtMe₃ | Good stability; suitable for noble metals | Lower volatility; longer pulse times needed; higher source temperatures |
| Metal β-diketonates | Pd(hfac)₂, Cu(hfac)₂ | Air-stable; easier handling | Low reactivity; fluorine incorporation from hfac ligand |
| Aminosilanes | BDEAS, 3DMAS, BTBAS | Designed for SiO₂ / SiNₓ ALD; non-pyrophoric | Require plasma co-reactant for SiO₂; slower GPC than metal oxides |
Co-Reactant Selection Guide
- H₂O: Most common oxidant for thermal ALD. Excellent for Al₂O₃, HfO₂, ZnO. Long purge times required due to strong surface affinity.
- O₃ (ozone): Stronger oxidant than H₂O. Shorter purge times, better nucleation on hydrophobic surfaces (e.g., H-terminated Si, noble metals). Preferred for batch and spatial ALD systems.
- O₂ plasma: Generates atomic oxygen radicals (O*). Essential for PEALD-SiO₂, PEALD metals, and low-temperature processes. Short purge times.
- NH₃: Thermal nitrogen source for nitrides. Limited reactivity, typically requires temperatures >300 °C.
- N₂/H₂ plasma: Generates NHₓ* radicals. Preferred for PEALD nitrides (TiN, TaN, SiNₓ, AlN, GaN) at lower temperatures with lower carbon and oxygen contamination.
- H₂ plasma: Reducing agent for metal ALD (W, Cu). Also used for ALD of pure elemental films.
5) Key Applications of ALD
The unique capabilities of ALD—angstrom-level thickness control, exceptional conformality, and pinhole-free coverage—make it the deposition method of choice for an expanding range of applications.
5.1 Semiconductor Gate Dielectrics & High-κ Films
ALD’s most transformative impact has been in semiconductor gate stack engineering. As transistor dimensions scaled below 45 nm, SiO₂ gate dielectrics became too thin (<2 nm) to prevent unacceptable tunneling leakage currents. ALD-grown HfO₂ (and HfSiOₓ, HfAlOₓ variants) replaced SiO₂ as the gate dielectric, enabling continued scaling with lower leakage. ALD is the only technique capable of depositing these 1–3 nm films with the required thickness uniformity (<1%), conformality in FinFET and gate-all-around (GAA) architectures, and interface quality. Today, ALD HfO₂ is also used for ferroelectric HfZrOₓ in emerging FeRAM and FeFET memories.
5.2 MEMS Passivation & Encapsulation
MEMS devices require ultra-thin, conformal passivation layers to protect delicate mechanical structures from moisture, oxidation, and stiction. ALD Al₂O₃ (10–50 nm) provides an excellent moisture barrier with pinhole-free coverage over released MEMS structures. The low deposition temperature of PEALD (<150 °C) is compatible with post-release MEMS processing, and ALD’s conformality ensures that undercuts, re-entrant features, and narrow gaps are uniformly coated.
5.3 Energy Devices
- Lithium-ion batteries: ALD Al₂O₃ coatings (1–5 nm) on cathode particles suppress parasitic reactions with the electrolyte, dramatically improving capacity retention and cycle life. ALD TiO₂ and ZnO coatings on anodes reduce solid-electrolyte interphase (SEI) growth.
- Solid-state batteries: ALD enables deposition of ultra-thin solid electrolyte layers (Li₃PO₄, LiAlO₂) with the pinhole-free coverage required for leak-free ionic conduction.
- Solar cells: ALD Al₂O₃ provides excellent surface passivation for PERC and TOPCon silicon solar cells, reducing surface recombination velocity to <5 cm/s. ALD TiO₂ is used as an electron transport layer in perovskite solar cells.
- Fuel cells & electrolysis: ALD Pt and Pd enable ultra-low-loading catalytic layers with high utilization efficiency on 3D electrode structures.
5.4 Optical Coatings & Photonics
ALD films exhibit extremely low surface roughness (<0.5 nm RMS) and precise thickness control, making them ideal for optical interference coatings. Applications include:
- Anti-reflection coatings: ALD Al₂O₃/TiO₂ multilayers on lenses and photodetectors
- Optical filters: Distributed Bragg reflectors (DBRs) from ALD-deposited high-/low-index pairs with angstrom-level period control
- Photonic crystals: ALD conformal coating of 3D photonic crystal templates to precisely tune the photonic bandgap
- Waveguide cladding: Low-loss ALD Al₂O₃ and HfO₂ cladding layers for integrated photonics
5.5 Protective & Functional Coatings
- Corrosion protection: ALD Al₂O₃ provides pinhole-free barrier coatings on metals, polymers, and 3D-printed parts
- Biomedical implants: ALD TiO₂ and ZnO coatings enhance biocompatibility and provide antimicrobial properties
- Diffusion barriers: ALD TiN and TaN serve as Cu diffusion barriers in BEOL interconnects with conformality that PVD cannot achieve in high-AR vias — for the downstream patterning of these barrier and gate-metal layers, see the Metal Etching Complete Guide
- Catalysis: ALD enables atomic-scale tuning of catalyst composition and overcoating to prevent sintering while maintaining activity
6) ALD vs Other Deposition Techniques
Understanding how ALD compares to alternative thin-film deposition methods is essential for selecting the right technique for each application. The following table provides a quantitative comparison across the most important performance metrics:

| Parameter | ALD | CVD | PVD (Sputtering) | PECVD | MBE |
|---|---|---|---|---|---|
| Thickness control | <1 Å (digital, per cycle) | ~5–10 nm (rate × time) | ~5–10 nm (rate × time) | ~5–10 nm (rate × time) | ~1 monolayer (RHEED control) |
| Step coverage (10:1 AR) | >98% | 50–80% | 10–30% (line-of-sight) | 40–70% | <10% (line-of-sight) |
| Uniformity (300 mm) | <1% 1σ | 2–5% | 2–5% | 2–5% | 1–3% (small wafers only) |
| Deposition rate | 0.5–2 Å/cycle (1–10 nm/min) | 10–100 nm/min | 10–100 nm/min | 10–200 nm/min | 0.1–1 nm/min (epitaxial) |
| Temperature range | 25–400 °C | 300–900 °C | RT–500 °C | 100–400 °C | 200–800 °C |
| Film density | Near bulk | Good | Good to excellent | Moderate (columnar voids) | Excellent (epitaxial) |
| Pinhole density | Extremely low | Low to moderate | Moderate (columnar growth) | Low to moderate | Very low |
| Composition control | Excellent (cycle ratio tuning) | Good (flow ratio control) | Limited (co-sputtering) | Good (flow ratio control) | Excellent (beam flux) |
| Throughput | Low (batch ALD improves this) | High | High | High | Very low |
| Best for | Ultra-thin films, conformal coatings, high-AR structures | Thick films, high throughput, epitaxial growth | Metals, hard coatings, optical films on flat substrates | Dielectric films, passivation, moderate conformality | Epitaxial single-crystal films, quantum structures |
When to choose ALD over alternatives: ALD is the preferred technique when any of the following requirements apply: (1) film thickness <20 nm with <5% tolerance, (2) conformal coating of structures with aspect ratios >5:1, (3) pinhole-free encapsulation at any thickness, (4) precise composition in ternary/quaternary alloys, or (5) deposition on temperature-sensitive substrates (with PEALD). For films thicker than ~100 nm on planar substrates, CVD or PVD will typically offer better throughput at lower cost per wafer.
7) Process Optimization & Troubleshooting
Even with ALD’s inherent self-limiting chemistry, achieving optimal film properties requires careful process development. This section provides a systematic approach to common ALD challenges.
7.1 Saturation Curve Characterization
The first step in any ALD process development is establishing the saturation curve: GPC as a function of precursor pulse time. Increase pulse duration incrementally while holding all other parameters constant. The GPC should plateau once the surface is fully saturated. Operating at the saturation plateau (plus a 20–30% margin) ensures true self-limiting growth.
- Undersaturated regime: GPC increases with pulse time. Film will be non-uniform (center-heavy or edge-heavy depending on gas distribution).
- Saturated regime: GPC constant with pulse time. Target operating regime.
- Oversaturated / CVD regime: GPC increases again due to precursor decomposition or condensation. Film properties degrade.
7.2 Purge Time Optimization
Insufficient purge time is the most common cause of degraded ALD film quality. If purge steps are too short, residual precursor or co-reactant remains in the chamber, leading to parasitic CVD reactions that destroy conformality and thickness control.
- H₂O purge: H₂O is notoriously difficult to purge due to its strong hydrogen-bonding affinity for oxide surfaces. Thermal ALD Al₂O₃ at 150 °C may require 15–30 s H₂O purge times. At 300 °C, 3–5 s is typically sufficient.
- Metal precursor purge: Most metal-organic precursors require 3–10 s purge times. Precursors with low vapor pressure (e.g., MeCpPtMe₃) may need longer purge with assist gas.
- Diagnostic: If GPC exceeds the expected saturation value and increases with pulse time beyond the saturation point, suspect insufficient purge. Double the purge time and re-measure.
7.3 Common ALD Problems & Solutions
| Problem | Possible Causes | Diagnostic | Solution |
|---|---|---|---|
| GPC higher than expected | CVD component from insufficient purge; precursor decomposition; precursor condensation | Run saturation curve; check if GPC varies with pulse time beyond saturation | Increase purge time; reduce substrate temperature (if decomposition); increase temperature (if condensation) |
| GPC lower than expected | Insufficient precursor dose; source temperature too low; blocked delivery line; substrate above ALD window | Check source temperature and cylinder level; run saturation curve | Increase pulse time or source temperature; verify delivery line heating; check for precursor depletion |
| Poor thickness uniformity | Undersaturation; non-uniform heating; precursor depletion across wafer | Map thickness (ellipsometry 49-point); compare center vs edge GPC | Increase pulse time to ensure full saturation; verify temperature uniformity; optimize showerhead gas distribution |
| High carbon content in film | Incomplete ligand removal; temperature too low; insufficient co-reactant exposure | XPS depth profile; compare films at different temperatures | Increase substrate temperature; increase co-reactant pulse time; switch to O₃ or plasma co-reactant |
| Particle contamination | Chamber wall flaking; gas-phase nucleation; precursor line condensation | Particle count (SPC chart); inspect chamber walls; check line temperatures | Clean chamber (NF₃ plasma or wet clean); increase line heating; add particle trap; verify purge adequacy |
| Nucleation delay | Substrate surface lacks reactive sites (e.g., H-terminated Si, noble metal surfaces) | Plot thickness vs cycle count; check for non-linear onset | Apply surface pretreatment (O₃ exposure, O₂ plasma, or UV-ozone); use O₃ instead of H₂O as co-reactant; apply seed layer |
| Poor step coverage | Insufficient precursor dose for high-AR features; recombination of plasma radicals on sidewalls (PEALD) | Cross-section TEM/SEM; compare top vs bottom thickness | Increase pulse time (may need 10× longer for extreme AR); switch from PEALD to thermal ALD; use multiple short pulses with intermediate purge |
7.4 Film Characterization Checklist
A comprehensive ALD process qualification should include the following measurements:
- Thickness & uniformity: Spectroscopic ellipsometry (SE) with 49-point or 81-point wafer map
- Refractive index: SE at 633 nm; compare to bulk reference values
- Composition & impurities: XPS, RBS, or SIMS depth profiling for C, N, Cl, H content
- Density: XRR (X-ray reflectometry) for film density and roughness
- Crystallinity: XRD or GIXRD for phase identification (amorphous, anatase, rutile, monoclinic, etc.)
- Electrical properties: CV and IV measurements for dielectric constant, breakdown field, leakage current density
- Step coverage: Cross-section SEM or TEM on patterned test structures
- Surface roughness: AFM (target <0.5 nm RMS for optical applications)
- Stress: Wafer bow measurement (Stoney equation) for compressive/tensile film stress
8) Equipment Selection Guide
Selecting the right ALD system requires matching the equipment capabilities to your process requirements. The following criteria should guide your evaluation:
8.1 Wafer Size & Throughput
ALD systems must be designed for the substrate sizes used in your application. Research and development labs may work with small coupons or 4″ wafers, while production fabs require 200 mm (8″) or 300 mm (12″) compatibility. For non-standard substrates (large panels, 3D objects, or powder batches), specialized “supersize” chamber configurations are available. Throughput can be enhanced through batch processing (multiple wafers per run) or spatial ALD architectures.
8.2 Temperature Range & Zones
The substrate heater must cover the full ALD temperature window for your target materials. A system supporting 20–400 °C covers the vast majority of ALD processes. Equally important are the precursor source temperatures (typically 20–150 °C, with 200 °C capability for low-volatility precursors) and delivery line heating to prevent condensation between the source and the chamber. Multi-zone temperature control ensures uniform heating across the substrate.
8.3 Vacuum & Gas Delivery
- Base pressure: A base pressure of <5×10⁻⁵ Torr ensures low background contamination and is essential for high-purity film growth. This requires a turbomolecular pump backed by a dry pump.
- Precursor lines: The number of independent precursor delivery lines determines process flexibility. Two lines suffice for a single binary ALD process; 4–6 lines enable multi-material deposition, in-situ multilayers, and alloy composition tuning without breaking vacuum.
- Gas distribution: Showerhead-type gas feed-in provides the most uniform precursor distribution across the wafer surface, essential for achieving <1% thickness uniformity.
8.4 Plasma Capability
If your application requires metal films, nitrides, or low-temperature deposition, a PEALD-capable system with remote plasma is essential. Remote plasma RF sources in the 300–1000 W range provide sufficient radical flux for most PEALD processes while minimizing substrate damage. Ensure the plasma source can operate with O₂, N₂, H₂, NH₃, and Ar/N₂ mixtures.
8.5 Chamber Design
Key chamber design features to evaluate:
- Box-in-box chamber architecture: An inner reaction chamber nested within an outer vacuum shell minimizes the chamber volume exposed to precursors, reducing purge times and precursor consumption while preventing parasitic deposition on chamber walls.
- Compact uni-body design: A single integrated frame simplifies installation, reduces the system footprint, and minimizes the number of vacuum seals and potential leak points.
- Chamber wall temperature control: Heated chamber walls (matched to substrate temperature or slightly above) prevent precursor condensation on walls, reducing particle generation and improving purge efficiency.
- In-situ monitoring ports: Optical ports for spectroscopic ellipsometry, QCM (quartz crystal microbalance), or residual gas analysis (RGA) enable real-time process monitoring and endpoint detection.
8.6 NineScrolls ALD System Specifications
| Specification | Value |
|---|---|
| Wafer sizes | 4″, 6″, 8″, 12″, or supersize |
| Substrate temperature | 20–400 °C |
| Source temperatures | 20–150 °C (up to 200 °C optional) |
| Base pressure | <5×10⁻⁵ Torr |
| Growth rate | 0.5–2 Å per cycle |
| Film uniformity | <1% (Al₂O₃) |
| Step coverage | >98% |
| Precursor lines | 2–6 |
| Remote plasma RF (optional) | 300–1000 W |
| Gas distribution | Showerhead gas feed-in |
| Chamber design | Compact uni-body, box-in-box chamber |
| Depositable materials | Al₂O₃, HfO₂, SiO₂, TiO₂, Ga₂O₃, ZnO, TiN, TaN, SiNₓ, AlN, GaN, Pt, Pd, W, Ru |
9) Frequently Asked Questions
What is the difference between ALD and CVD?
The fundamental difference is that ALD uses sequential, self-limiting surface reactions, while CVD relies on continuous, simultaneous precursor flow. In CVD, both the metal precursor and the reactant gas are present in the chamber at the same time, leading to continuous deposition controlled by gas-phase transport and surface kinetics. In ALD, precursors are introduced one at a time with purge steps in between, so the surface chemistry self-terminates after each half-reaction. This gives ALD superior thickness control, conformality, and uniformity, but at the cost of lower throughput. Use ALD when you need films <50 nm with strict uniformity, conformality on 3D structures, or pinhole-free coverage. Use CVD when throughput matters and the substrate is relatively planar.
How long does a typical ALD process take?
A single ALD cycle (precursor pulse + purge + co-reactant pulse + purge) typically takes 2–30 seconds depending on the material system and temperature. Thermal ALD Al₂O₃ at 200 °C is among the fastest processes at ~4 s/cycle with ~1.1 Å/cycle GPC, yielding ~10 nm in about 6 minutes (~90 cycles). Processes requiring long H₂O purges at low temperature, or noble metal PEALD processes with extended plasma steps, can take 15–30 s/cycle. For a 20 nm Al₂O₃ film (~180 cycles), expect roughly 12–15 minutes of deposition time plus chamber conditioning overhead.
Can ALD deposit metals?
Yes. PEALD is the primary route for depositing metallic films by ALD. Noble metals such as Pt, Pd, and Ru are deposited using organometallic precursors with O₂ plasma or O₃ co-reactant. Conductive nitrides like TiN and TaN (which behave as metals electrically) are deposited using amide precursors with N₂/H₂ plasma. Tungsten (W) can be deposited using WF₆ with Si₂H₆ (thermal) or H₂ plasma. Thermal metal ALD is more limited due to the difficulty of reducing metal precursors without plasma assistance. The nucleation phase of metal ALD on oxide surfaces can be challenging (island growth), so seed layers or surface pretreatments are often required for continuous ultra-thin metal films.
What is the minimum film thickness achievable with ALD?
ALD can deposit films as thin as a single sub-monolayer (~0.5–1 Å, i.e., one cycle). However, at these thicknesses, the film may not be continuous due to nucleation effects, especially on non-ideal surfaces. For most practical applications, ALD films become continuous and pinhole-free at around 2–5 nm. For gate dielectrics, functional HfO₂ films as thin as 1 nm have been demonstrated. The minimum useful thickness depends on the material, substrate surface chemistry, and the specific application requirements.
How do I choose between thermal ALD and PEALD?
Use thermal ALD when: (1) you need the highest possible step coverage in extreme-aspect-ratio structures (>50:1), (2) the substrate is sensitive to plasma damage (e.g., organic electronics, OLED), (3) you are depositing standard oxides (Al₂O₃, HfO₂, ZnO) at temperatures above 150 °C, or (4) simplicity and lower system cost are priorities. Use PEALD when: (1) you need low-temperature deposition (<150 °C), (2) you are depositing metals (Pt, Pd, Ru, W) or nitrides (TiN, TaN, SiNₓ, AlN, GaN), (3) you need higher film density and lower impurity levels at a given temperature, or (4) you require shorter cycle times. Many labs choose systems with optional remote plasma capability to have maximum process flexibility.
What is the ALD temperature window and why does it matter?
The ALD temperature window is the range of substrate temperatures within which the growth per cycle (GPC) remains constant and the deposition is truly self-limiting. Below this window, precursor condensation or incomplete reactions occur; above it, precursor decomposition or desorption disrupts the self-limiting mechanism. Operating within the ALD window is essential for achieving the characteristic benefits of ALD: digital thickness control, excellent uniformity, and high conformality. For the workhorse TMA/H₂O Al₂O₃ process, the ALD window spans approximately 100–350 °C. Each precursor-reactant combination has its own characteristic window, which must be determined experimentally during process development.
How many precursor lines do I need?
The number of precursor lines depends on your process requirements. 2 lines suffice for a single binary ALD process (e.g., one metal precursor + one oxidant). 3–4 lines enable ternary alloys (e.g., HfAlOₓ using both TDMAH and TMA), in-situ bilayers (e.g., Al₂O₃/HfO₂ nanolaminate), or switching between thermal and plasma co-reactants. 5–6 lines provide maximum flexibility for multi-material stacks, quaternary compositions, and rapid process development without reconfiguring the gas panel. If you anticipate your research evolving toward new materials over the system’s lifetime, investing in additional precursor lines upfront is strongly recommended—adding lines later is significantly more expensive.
Is ALD suitable for production, or only R&D?
ALD is used extensively in high-volume manufacturing. Every advanced logic chip (Intel, TSMC, Samsung) uses ALD for gate dielectric deposition, and 3D NAND flash memory relies on ALD for charge-trap and blocking oxide layers at every word line. Production ALD throughput challenges are addressed through batch ALD (processing 50–200 wafers simultaneously in a vertical furnace), spatial ALD (separating precursor zones physically rather than temporally for continuous wafer processing), and optimized single-wafer systems with rapid cycling (<3 s/cycle). For R&D, single-wafer systems provide maximum process flexibility and fast recipe iteration. The NineScrolls ALD platform is designed to serve both R&D process development and pilot-scale production needs.
What maintenance does an ALD system require?
Routine ALD maintenance includes: (1) Chamber cleaning every 500–2000 cycles (or when particle counts increase) using in-situ NF₃ plasma clean or periodic wet clean with dilute HF or NaOH depending on the deposited material. (2) Precursor source replacement when cylinder pressure drops below the minimum operating threshold. (3) Pump maintenance per manufacturer schedule (oil changes for rotary vane pumps; bearing replacement for dry pumps and turbo pumps). (4) Delivery line inspection for condensation or blockage, especially after precursor changes. (5) O-ring and seal replacement annually or when leak rate increases above specification. Establishing a preventive maintenance schedule based on cycle count tracking significantly extends system uptime and film quality consistency.
What safety precautions are needed for ALD precursors?
ALD precursor safety is paramount. Pyrophoric precursors (TMA, DEZ) ignite spontaneously on contact with air and require inert-atmosphere glove-box handling, self-sealing cylinder connections, and a nitrogen-purged gas cabinet with fire suppression. Corrosive precursors (TiCl₄, HfCl₄, WF₆) produce HCl or HF by-products that require corrosion-resistant exhaust lines (316L stainless steel or Hastelloy) and a scrubbed exhaust system. Toxic precursors require gas monitoring with automatic shutoff interlocks. All ALD installations should include: an exhaust-ventilated gas cabinet, toxic gas monitoring, an emergency shutoff system, proper PPE (fire-resistant gloves for pyrophorics, acid-resistant gloves for halides), and operator training in MSDS procedures for each precursor in use.
NineScrolls ALD Systems
Our ALD systems feature a compact uni-body design with box-in-box chamber architecture, showerhead gas feed-in, and optional remote plasma (300–1000 W) for both thermal ALD and PEALD. Supporting wafer sizes from 4″ to 12″ and beyond, with 2–6 precursor lines and >98% step coverage, the NineScrolls ALD platform is engineered for research flexibility and production-grade film quality.