Process Chamber Materials & Contamination Control in Semiconductor Equipment
By NineScrolls Engineering · 2026-03-25 · 13 min read · Materials Science
Target Readers: Process engineers, equipment engineers, facilities/maintenance engineers, lab managers, and procurement teams responsible for specifying, operating, and maintaining semiconductor processing equipment (plasma etchers, CVD systems, plasma cleaners). Engineers troubleshooting particle issues, process drift, or contamination-related yield loss will find the diagnostics and cleaning protocol sections especially relevant.
TL;DR Summary
The materials that make up a process chamber — body, electrodes, liners, seals, and gas delivery components — directly influence film purity, particle performance, process repeatability, and maintenance cost. This guide covers the selection rationale for chamber body materials (aluminum alloys, stainless steel, ceramics), surface treatments (hard anodization, yttria coating, electropolishing), electrode design, liner and shield strategies, seal materials for various chemistries, and gas delivery material requirements. It then addresses the five major contamination sources (particles, metallic, organic, moisture, and cross-contamination) and provides actionable protocols for chamber conditioning (seasoning), preventive maintenance, cleaning (wet, plasma, mechanical), and post-maintenance qualification testing — the complete toolkit for maintaining contamination-free semiconductor processing.
1) Why Chamber Materials Matter
In any plasma process — whether reactive ion etching, PECVD deposition, or plasma cleaning — the process chamber interior is a chemically active environment. Energetic ions, reactive radicals, UV photons, and elevated temperatures interact with every exposed surface. The chamber materials must therefore satisfy a demanding set of requirements:
- Chemical inertness: Resist attack by process gases (fluorine, chlorine, oxygen radicals) and etch byproducts
- Low particle generation: Withstand plasma bombardment without sputtering, flaking, or shedding particles onto the wafer
- Low outgassing: Minimize release of water vapor, hydrocarbons, and dissolved gases that contaminate the process environment
- Thermal stability: Maintain structural integrity and dimensional stability across the operating temperature range
- Electrical properties: Provide appropriate conductivity (electrodes) or insulation (chamber walls, RF windows) as the design requires
- Cleanability: Allow effective removal of process deposits through wet chemical, plasma, or mechanical cleaning without surface degradation
Material choices involve trade-offs. Aluminum is lightweight and thermally conductive but corrodes in chlorine plasmas. Stainless steel is robust but heavy and a potential source of metallic contamination (Fe, Cr, Ni). Ceramics are plasma-resistant but brittle and expensive. Understanding these trade-offs enables engineers to make informed decisions when specifying equipment and planning maintenance strategies.
2) Chamber Body Materials
| Material | Advantages | Limitations | Surface Treatment | Best Suited For |
|---|---|---|---|---|
| 6061-T6 Aluminum | Lightweight, excellent thermal conductivity, easy machining, low cost | Corroded by Cl₂/BCl₃ without protection; soft (scratches easily) | Hard anodization (Type III), Y₂O₃ spray coating | Most etch and CVD process chambers (with anodization) |
| 304/316L Stainless Steel | High strength, weldable, good corrosion resistance, UHV-compatible | Heavy, metallic contamination risk (Fe, Cr, Ni), lower thermal conductivity | Electropolishing, passivation | UHV chambers, gas delivery, flanges, non-plasma-exposed structural components |
| Alumina (Al₂O₃) Ceramic | Excellent plasma resistance, electrically insulating, low sputtering yield | Brittle, expensive, difficult to machine complex shapes | Glazing, polishing | Plasma-facing chamber liners, RF windows, insulator rings |
| Quartz (Fused Silica) | High purity, UV-transparent, good for fluorine-based processes | Etched by fluorine plasmas (consumable), thermal shock risk | None (inherently pure) | Process tube liners, ICP dielectric windows, RIE chamber liners |
| Silicon Carbide (SiC) | Extreme hardness, thermal shock resistance, good plasma resistance | Very expensive, limited machinability | CVD coating on graphite substrate | Susceptors, high-temperature chamber components |
2.1 Aluminum — The Dominant Chamber Material
Aluminum alloy (typically 6061-T6) is the most widely used chamber body material in semiconductor processing equipment. Its combination of low weight, excellent thermal conductivity (167 W/m·K), easy machinability, and moderate cost makes it the default choice for RIE, ICP-RIE, PECVD, and plasma cleaning chambers. However, bare aluminum is rapidly attacked by chlorine- and bromine-containing plasmas and corrodes in humid conditions. Surface treatment is therefore essential.
2.2 Stainless Steel — Robustness for Non-Plasma Surfaces
316L stainless steel (low-carbon variant for improved weldability and corrosion resistance) is used for structural components, vacuum flanges, gas delivery lines, and chambers that require UHV capability or high-temperature bakeout. Its primary limitation as a plasma-facing material is the risk of sputtering Fe, Cr, and Ni atoms onto the wafer — metallic contaminants that create deep-level traps in silicon, degrading device performance. For this reason, stainless steel is generally avoided in direct plasma exposure and is used for the chamber exterior, foreline, and mechanical support structures.
3) Surface Treatments
Surface treatments transform the properties of base chamber materials, dramatically improving their plasma resistance, particle performance, and corrosion behavior.
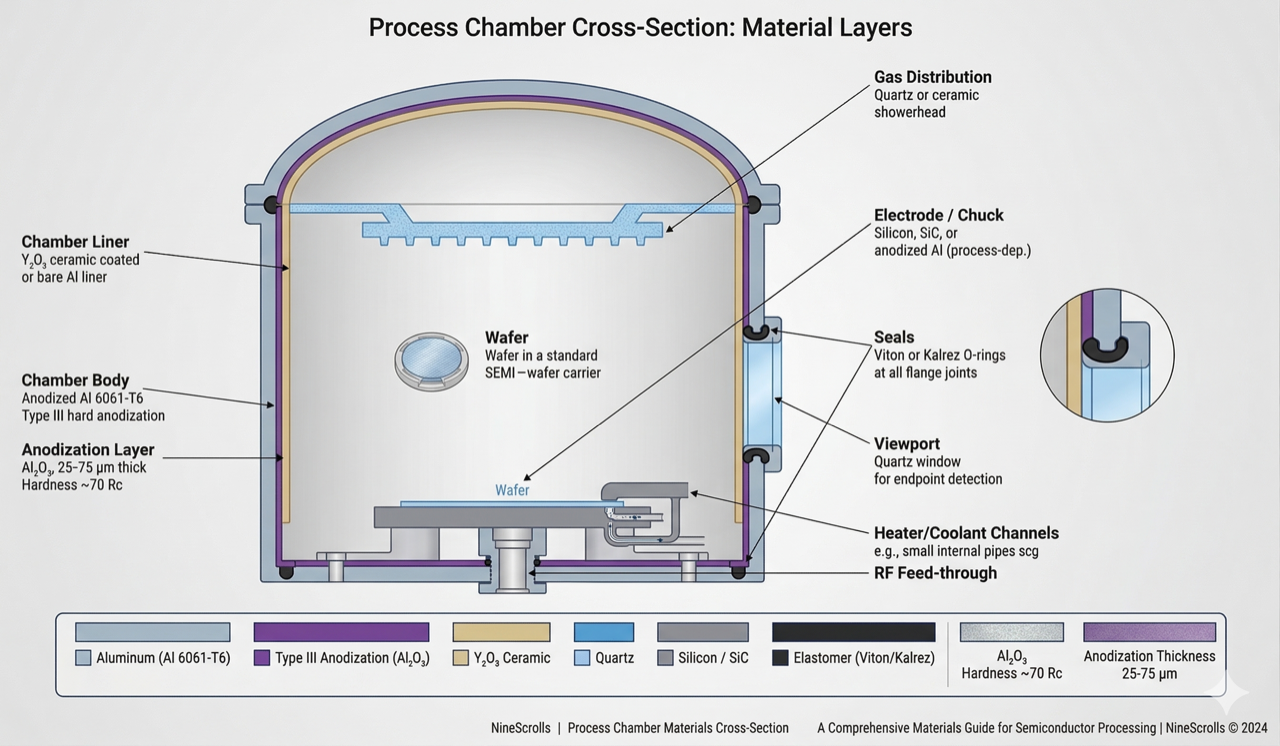
Figure 1: Chamber construction materials — cross-section showing anodized aluminum body, yttria ceramic liners, silicon or SiC electrode, quartz showerhead, and elastomer seals at each critical interface
3.1 Hard Anodization (Type III)
Hard anodization is the standard surface treatment for aluminum process chambers. The process electrochemically grows a dense Al₂O₃ layer (typically 25–75 µm thick) on the aluminum surface. This layer provides:
- Corrosion protection: The Al₂O₃ layer resists fluorine and chlorine plasma attack far better than bare aluminum
- Reduced particle shedding: The anodized surface is harder (Vickers hardness ~400–600) and more resistant to physical erosion from ion bombardment
- Electrical insulation: The anodized layer acts as a dielectric, which can be advantageous or problematic depending on RF coupling requirements
- Improved outgassing: Properly sealed anodization has lower outgassing than bare machined aluminum
Limitations: Anodization is not infinitely durable. Over thousands of plasma hours, the anodized layer erodes — particularly in fluorine-rich environments. Localized failure (pitting) exposes bare aluminum, leading to accelerated corrosion, particle generation, and metallic contamination. Monitoring anodization integrity is a key maintenance activity, and re-anodization is required periodically (typically every 1–3 years depending on process chemistry and chamber utilization).
3.2 Yttria (Y₂O₃) Coating
Yttrium oxide (Y₂O₃) plasma-sprayed coatings represent a significant advancement over standard anodization for aggressive plasma environments. Y₂O₃ has very low sputtering yield and excellent resistance to both fluorine and chlorine plasmas. Key advantages:
- 3–5× longer lifetime than hard anodization in fluorine-based etch processes
- Lower particle generation rates under sustained plasma exposure
- Reduced metallic contamination contribution compared to eroding anodization
- Available as plasma-sprayed coating (100–200 µm) on aluminum substrates
Y₂O₃ coatings are increasingly common in production etch equipment and represent a worthwhile upgrade for research tools that run high-volume fluorine-based processes. The coating cost premium over anodization is recovered through extended maintenance intervals and improved particle performance.
3.3 Electropolishing
Electropolishing removes the outermost layer of stainless steel through controlled anodic dissolution in an electrolyte bath, producing an ultra-smooth surface (Ra < 0.25 µm). Benefits for vacuum and semiconductor applications include:
- Reduced surface area → lower outgassing rate (critical for achieving UHV)
- Removal of embedded machining debris, tool marks, and surface inclusions
- Formation of a chromium-enriched passive layer that improves corrosion resistance
- Easier cleaning — smooth surfaces trap fewer particles and are more effectively cleaned
All high-quality stainless steel gas delivery tubing and fittings (316L EP) should be electropolished to semiconductor standards. Chamber components made of stainless steel benefit from electropolishing followed by passivation in hot nitric acid.
4) Electrode Materials
In plasma etch systems, the powered and grounded electrodes are among the most stressed components — subjected to intense ion bombardment, radical flux, and thermal cycling. Electrode material selection directly impacts etch uniformity, contamination levels, and electrode lifetime.
| Electrode Material | Typical Application | Advantages | Limitations |
|---|---|---|---|
| Anodized Aluminum | RIE lower electrode (wafer chuck) | Good thermal conductivity for wafer temperature control, economical | Anodization erosion, Al contamination risk if coating fails |
| Silicon (Si) | ICP-RIE upper electrode (showerhead), RIE electrodes for Si etch | No metallic contamination; consumed electrode contributes only Si (benign in Si processing) | Consumable (must be replaced periodically), expensive for large sizes |
| Silicon Carbide (SiC) | Electrode components in aggressive plasma environments | Very high erosion resistance, good thermal properties | Expensive, limited to simple geometries |
| Graphite (C) | Upper electrodes in some CCP-RIE systems | No metallic contamination, good RF conductivity | Generates particles, consumed in O₂-containing plasmas |
Silicon electrodes deserve special mention. Many modern etch systems use high-purity silicon for plasma-facing electrode surfaces. Since silicon is the primary substrate material being processed, any sputtered electrode material is "process-compatible" and does not introduce foreign metallic contamination. Silicon showerhead electrodes in ICP-RIE systems serve double duty as gas distribution plates and grounded electrodes, with hundreds of precisely machined holes for uniform gas delivery.
5) Liner and Shield Design
Chamber liners (also called shields or inserts) are removable components that line the interior walls of the process chamber, serving two critical functions:
- Protection: They protect the permanent chamber body from plasma erosion and process deposit buildup, extending the chamber body lifetime
- Simplified maintenance: When deposits accumulate to unacceptable levels, liners are removed for cleaning or replacement — far easier and cheaper than cleaning or refurbishing the entire chamber body
Liner Material Options
- Anodized aluminum liners: Standard in most etch systems. Moderate cost, good plasma resistance, and easy to manufacture. Replaced when anodization is compromised
- Y₂O₃-coated aluminum liners: Premium option for fluorine-heavy processes. Longer service life between cleanings
- Quartz liners: Used in some RIE and ICP-RIE systems, especially for fluorine-based silicon etching. The quartz acts as a benign, high-purity shield. The liner is consumed slowly and replaced periodically
- Ceramic liners (Al₂O₃): Used in high-power ICP systems where ion bombardment of the walls is intense. Excellent sputtering resistance
Design consideration: Liners must be thermally managed. If a liner runs too cold, process deposits adhere more tenaciously and can flake off as particles. If a liner runs too hot, certain deposits may not adhere at all, leading to loose particles. Some advanced chamber designs include active temperature control (heaters or coolant channels) for liner surfaces to optimize deposit adhesion and minimize particle generation.
6) O-Ring and Seal Materials
Vacuum seals are critical contamination barriers. The choice of seal material must account for the process chemistry, operating temperature, and required base pressure. See the comparison of common seal materials:
| Seal Material | Max Temp | Fluorine Resistance | Chlorine Resistance | Outgassing | Recommended Use |
|---|---|---|---|---|---|
| Viton (FKM) | 200°C | Good | Good | Low | Standard semiconductor O-ring; F₂ and Cl₂ chemistries at moderate temperatures |
| Kalrez (FFKM) | 315°C | Excellent | Excellent | Very low | Aggressive chemistries (NF₃, ClF₃), high-temperature processes, critical contamination control |
| Chemraz (FFKM) | 300°C | Excellent | Excellent | Very low | Alternative to Kalrez; similar performance at slightly lower cost |
| Silicone (VMQ) | 230°C | Poor | Poor | High | Not recommended for semiconductor process chambers; acceptable for non-critical rough vacuum |
| PTFE (Teflon) | 260°C | Excellent | Excellent | Very low | Gas delivery seals, static seals (not suitable for high-compression vacuum seals due to cold flow) |
| Metal Seals (Cu, Ni, Al) | > 450°C | Excellent | Excellent | Negligible | UHV flanges, high-temperature joints, bakeout-compatible connections |
O-ring maintenance best practice: During every chamber opening, inspect all accessible O-rings for cracks, flat spots (compression set), discoloration, and surface roughness. Clean the O-ring groove with a lint-free wipe and isopropyl alcohol. Apply a thin, even layer of vacuum-grade fluorinated grease (e.g., Krytox or Fomblin) to the O-ring — enough to produce a visible sheen but not so much that excess grease enters the vacuum system. Replace O-rings at the manufacturer's recommended interval or when any sign of degradation is observed.
7) Gas Delivery Materials
The gas delivery system — from gas cabinet to mass flow controller to chamber inlet — must maintain gas purity from source to process. Contamination introduced by gas delivery components (particles, moisture, metallic ions) directly affects process results.
- Tubing: 316L stainless steel, electropolished (EP) to Ra < 0.25 µm internal finish, is the standard for semiconductor gas delivery. EP tubing minimizes particle generation, moisture adsorption, and corrosion by process gases
- Fittings: Face-seal fittings (VCR type) with metal gaskets provide leak-tight, particle-free connections. Compression fittings (Swagelok type) are acceptable for non-critical lines but are less reliable for ultra-high-purity applications
- Regulators and valves: All-metal diaphragm valves with 316L EP wetted surfaces. Avoid elastomer-seated valves in corrosive gas lines (fluorine, chlorine)
- Purge capability: Gas lines should be designed for purge/pump-out to remove moisture and atmospheric contamination before process gas introduction. Dead legs (sections of tubing that cannot be swept by gas flow) must be minimized
- PTFE components: PTFE (or PFA) tubing and fittings are used for certain specialty gases and downstream of the chamber (exhaust side) where chemical resistance is prioritized over ultra-low particle generation
8) Contamination Sources and Classification
Contamination in semiconductor process chambers falls into five categories, each with distinct sources, detection methods, and mitigation strategies:
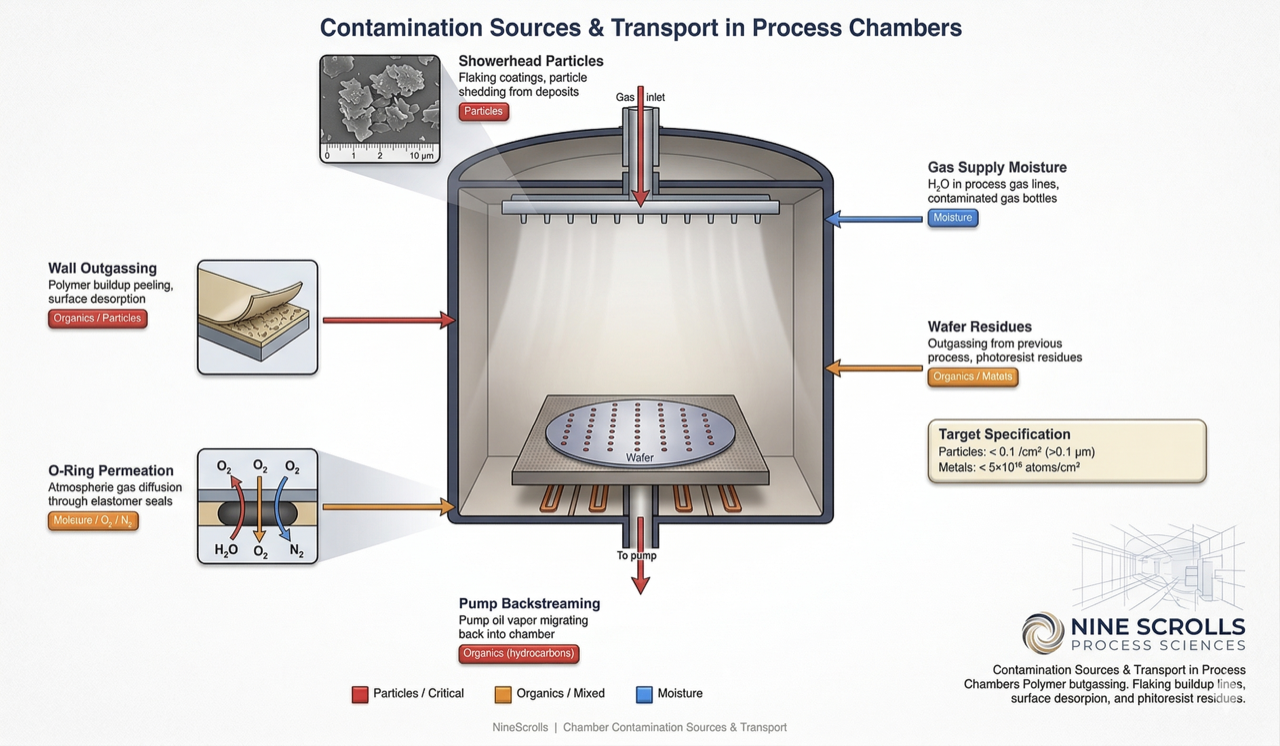
Figure 2: Contamination sources — six major pathways for particles, moisture, organics, and metal contamination entering the process environment, from showerhead flaking to pump oil backstreaming
| Contamination Type | Sources | Impact on Process | Detection Method |
|---|---|---|---|
| Particles | Flaking deposits, eroded chamber surfaces, O-ring debris, process byproduct nucleation | Pattern defects, short circuits, etch non-uniformity | Wafer surface scanner (KLA Surfscan), optical inspection |
| Metallic | Sputtered chamber/electrode material (Fe, Cr, Ni, Al), gas impurities, corroded fittings | Deep-level traps in Si (lifetime degradation), gate oxide reliability failure | TXRF, VPD-ICP-MS, SIMS |
| Organic | O-ring outgassing, pump oil backstreaming, photoresist residue, fingerprints | Poor film adhesion, resist scumming, contact resistance increase | Contact angle measurement, ATR-FTIR, TOF-SIMS |
| Moisture (H₂O) | Chamber venting, leaks, O-ring permeation, gas line contamination | Oxidation of sensitive surfaces, etch rate instability, film composition drift | RGA (mass 18), moisture sensor, base pressure trend |
| Cross-contamination | Previous process residues (e.g., Cl from metal etch contaminating oxide etch) | Process drift, unexpected etch chemistry, device failure | Dummy wafer test + SIMS/XPS, process trending |
Particle management is typically the most visible and closely tracked contamination metric. Modern semiconductor fabs specify particle "adder" limits per wafer pass (e.g., < 10 particles ≥ 0.1 µm per 200 mm wafer). Research labs may accept higher counts but should still track particle trends to catch degradation before it affects experimental results.
9) Chamber Conditioning (Seasoning)
After any maintenance event that exposes the chamber interior to atmosphere — liner replacement, electrode change, wet cleaning — the chamber must be re-conditioned before running product wafers. Chamber conditioning (also called "seasoning") establishes a stable, reproducible surface state on all plasma-facing components.
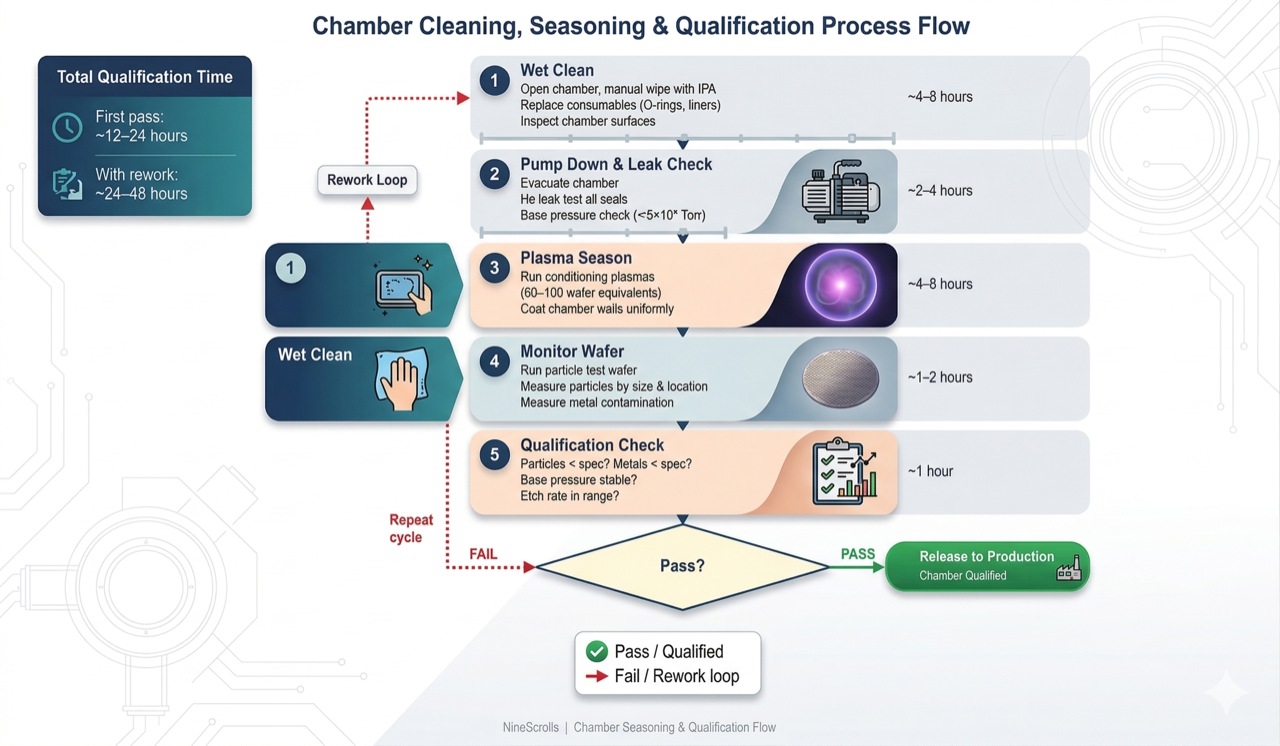
Figure 3: Chamber conditioning workflow — five-stage process from wet clean through plasma seasoning to qualification, with pass/fail decision gate and recycle loop for out-of-spec results
Seasoning Process
- Pump-down and leak check: Pump the chamber to base pressure, verify leak rate meets specification (He leak test or rate-of-rise)
- Bakeout (optional but recommended): Heat chamber walls to 80–120°C for 2–4 hours to accelerate water desorption
- Plasma conditioning: Run a series of plasma steps to sputter-clean electrode surfaces, deposit a controlled coating on chamber walls, and stabilize surface chemistry. Typical seasoning recipes include:
- O₂ plasma (5–15 min) to remove organic residues
- Process-gas plasma (e.g., CF₄/O₂ for oxide etch chambers, Cl₂/BCl₃ for metal etch) to establish the "working" chamber surface state
- Multiple dummy wafer runs (5–25 wafers) using the production recipe to reach steady-state conditions
- Qualification testing: Run particle monitor wafers and process qualification wafers to verify the chamber meets specifications before returning to production
The number of seasoning wafers required varies by process and chamber design. A well-designed chamber with Y₂O₃-coated liners may reach steady state in 5–10 dummy wafers, while a bare anodized aluminum chamber running a polymer-depositing etch process may need 20–30 wafers. Tracking the etch rate (or deposition rate) trend vs. wafer count during seasoning provides an objective metric for determining when the chamber is ready for production.
10) Preventive Maintenance Schedules
A structured preventive maintenance (PM) program prevents unexpected downtime, maintains particle and process performance, and extends equipment lifetime. The following table provides typical PM intervals; actual intervals should be adjusted based on process chemistry, utilization, and observed performance trends.
| PM Level | Frequency | Tasks | Downtime |
|---|---|---|---|
| In-situ Clean | Every wafer or every N wafers | Plasma clean between wafers (O₂, NF₃, or CF₄ plasma) to remove process deposits | 30 s – 5 min (automated) |
| Light PM | Every 500–2,000 RF hours | O-ring inspection, viewport cleaning, particle monitor wafer, foreline trap check | 2–4 hours |
| Medium PM | Every 2,000–5,000 RF hours | Liner removal and cleaning (or replacement), O-ring replacement, electrode inspection, full chamber wipe-down, leak check, re-seasoning | 8–24 hours |
| Heavy PM | Every 5,000–15,000 RF hours | Complete chamber disassembly, electrode replacement, liner re-anodization or replacement, turbo pump service, full wet clean of chamber body, helium leak test, extended re-seasoning | 2–5 days |
11) Cleaning Protocols
Chamber cleaning removes accumulated process deposits, particles, and contamination. Three primary cleaning methods are used, often in combination:
11.1 Plasma Cleaning (In-Situ)
Plasma cleaning uses reactive gas plasmas to volatilize and remove deposits without opening the chamber. It is the first line of defense against contamination buildup. For detailed guidance, see our plasma cleaner maintenance guide.
- O₂ plasma: Removes organic deposits (photoresist, polymer residue) by forming volatile CO, CO₂, and H₂O. Standard inter-wafer clean for etch processes that deposit fluorocarbon polymers
- NF₃ or CF₄ plasma: Removes silicon-containing deposits (SiO₂, SiN, a-Si) through fluorine radical attack. Used after CVD processes and for periodic deep cleaning of etch chambers
- Ar plasma: Physical sputtering of non-volatile deposits. Less selective but effective for metallic contamination removal
11.2 Wet Cleaning (Ex-Situ)
Wet cleaning involves removing chamber components (liners, shields, electrodes) and cleaning them with chemical solutions outside the chamber. Typical wet clean procedures:
- Degreasing: Ultrasonic cleaning in acetone or isopropyl alcohol to remove organic contamination
- Acid cleaning: Immersion in dilute HF (for oxide removal), dilute HNO₃ (for metallic contamination removal), or proprietary semiconductor-grade cleaning solutions
- DI water rinse: Multiple rinse cycles in deionized water (18.2 MΩ·cm) to remove acid residues
- Drying: Nitrogen blow-dry or oven dry at 80–120°C. Avoid air drying, which leaves water spots and re-adsorbs moisture
- Inspection: Visual inspection under bright light for residual contamination, surface damage, or anodization failure
11.3 Mechanical Cleaning (Bead Blasting)
Bead blasting (using alumina or glass bead media) is used to remove heavily adhered deposits that resist plasma and wet chemical cleaning. It is typically applied to liners and shields during heavy PM events. Important considerations:
- Use only media approved for semiconductor applications (high-purity alumina beads, typically 50–100 µm)
- Bead blasting roughens the surface, which improves deposit adhesion (reducing flaking/particle generation) but increases surface area (potentially increasing outgassing)
- After bead blasting, components must be thoroughly ultrasonically cleaned and rinsed to remove embedded media particles
- Do not bead-blast anodized surfaces unless re-anodization is planned — the process destroys the anodized layer
12) Qualification Criteria
After any maintenance event that disturbs the process chamber, the system must be re-qualified before processing product wafers. Qualification verifies that the chamber meets its performance specifications and is free from contamination.
Standard Qualification Tests
| Test | What It Measures | Typical Acceptance Criteria | Method |
|---|---|---|---|
| Base Pressure | Vacuum integrity, outgassing level | ≤ specified base pressure within specified pump-down time | Monitor chamber pressure after pump-down |
| Leak Rate (He test) | Seal integrity, weld quality | < 1 × 10⁻⁹ Torr·L/s (production); < 5 × 10⁻⁹ (research) | He mass spectrometer leak detector |
| Rate-of-Rise | Combined leak + outgassing rate | < 2 mTorr/min (typical for anodized Al chamber) | Isolate chamber, monitor pressure increase |
| Particle Count | Particle generation from chamber surfaces | < 10–30 adders ≥ 0.1 µm per wafer (process-dependent) | Run bare Si monitor wafer through process recipe, measure pre/post particle count |
| Dummy Wafer Test | Process performance (rate, uniformity, selectivity) | Etch/deposition rate within ±5% of baseline; uniformity within spec | Process oxide/nitride/Si wafer, measure with ellipsometer or profilometer |
| Metallic Contamination | Surface metal contamination from chamber materials | < 10¹⁰ atoms/cm² for critical metals (Fe, Cu, Ni, Cr) | VPD-ICP-MS or TXRF on bare Si monitor wafer |
Qualification sequence: The recommended order is: (1) base pressure verification, (2) leak test, (3) chamber seasoning (5–25 dummy wafers), (4) particle monitor wafer, (5) process qualification wafer. If any step fails, diagnose and correct before proceeding. Do not attempt to "season through" a leak or base pressure problem — these must be resolved first.
For additional context on how chamber condition affects plasma processes, see our guides on plasma etching fundamentals and plasma cleaner maintenance.
NineScrolls Process Equipment — Engineered for Contamination Control
Our etch, CVD, and plasma cleaning systems feature hard-anodized or Y₂O₃-coated chamber surfaces, silicon or SiC electrodes, replaceable chamber liners, automated in-situ plasma cleaning, and Kalrez-sealed load-locks — designed to minimize particle generation and maximize time between maintenance events.
Frequently Asked Questions
Why is anodized aluminum preferred over stainless steel for plasma etch chamber bodies?
Anodized aluminum offers several advantages over stainless steel for plasma-facing chamber surfaces. First, aluminum has far superior thermal conductivity (167 W/m·K vs. ~16 W/m·K for SS316), enabling better temperature uniformity across the chamber walls — important for consistent process deposit adhesion and etch uniformity. Second, the hard anodization layer (Al₂O₃) is chemically similar to the sapphire and alumina ceramics used in other plasma-resistant components, providing good resistance to fluorine and chlorine radical attack. Third, when an anodized aluminum surface does erode, it releases aluminum and oxygen — elements that are generally less harmful to silicon device performance than the iron, chromium, and nickel released from sputtered stainless steel. Finally, aluminum is significantly lighter and easier to machine, reducing equipment weight and manufacturing cost. The main limitation is that anodization degrades over time in aggressive plasma environments and must be periodically refurbished, whereas stainless steel (while a worse choice for plasma-facing surfaces) is inherently more mechanically robust.
How often should chamber liners be replaced, and what are the signs that replacement is needed?
Liner replacement intervals depend heavily on process chemistry, RF power, and throughput. As a general guideline, anodized aluminum liners in a fluorine-based etch process (SF₆/CF₄/C₄F₈) typically last 2,000–5,000 RF hours before the anodization is significantly eroded. Y₂O₃-coated liners may last 5,000–15,000 RF hours under similar conditions. Key indicators that liner replacement is needed include: (1) rising particle counts on monitor wafers (the most sensitive early indicator); (2) visible discoloration, pitting, or flaking on the liner surface when inspected during PM; (3) detection of increasing aluminum (or other metals) on dummy wafer surfaces via TXRF or VPD-ICP-MS; (4) process drift (etch rate or uniformity changes) that does not respond to recipe adjustment; and (5) the liner surface has lost its original anodized color and shows bare aluminum through the coating. Waiting until multiple indicators are present risks particle excursions on product wafers, so best practice is to establish trending thresholds and replace liners proactively when the first indicator triggers.
What is chamber seasoning and why can't I skip it after maintenance?
Chamber seasoning is the process of running a series of plasma conditioning steps and dummy wafers after maintenance to establish a stable, reproducible surface state on all chamber components before processing product wafers. It cannot be skipped because a freshly cleaned or newly installed chamber surface has a fundamentally different chemical state than a "working" chamber: surfaces are rich in adsorbed water, have no protective polymer coating (in etch processes that form sidewall passivation), and have different radical recombination coefficients. The first several wafers processed in an unseasoned chamber will exhibit different etch rates, selectivities, and profiles compared to steady-state conditions — this variation can cause yield loss on product wafers. During seasoning, the chamber walls accumulate a thin layer of process deposits that stabilizes radical concentration, wall recombination rates, and particle adhesion. By running 5–25 dummy wafers (depending on the process), you ensure that the chamber has reached this steady state before introducing valuable product wafers. Skipping seasoning to save time typically costs more in yield loss and re-work than the dummy wafers and time invested.
When should I use Kalrez O-rings instead of standard Viton?
Kalrez (FFKM perfluoroelastomer) O-rings should be used instead of Viton (FKM fluoroelastomer) in the following situations: (1) the process involves highly aggressive chemistries such as NF₃ remote plasma clean, ClF₃ chamber clean, or concentrated fluorine/chlorine gas exposure at the seal location — Kalrez has significantly better chemical resistance to these species; (2) the seal operates at elevated temperatures above 200°C, which exceeds Viton's reliable operating range — Kalrez is rated to 315°C; (3) the application requires the lowest possible outgassing, such as seals on UHV-compatible chambers or transfer modules; and (4) you are experiencing unexpectedly short Viton O-ring lifetimes (less than 3–6 months) due to chemical attack, indicated by O-rings that become hard, cracked, swollen, or sticky. Kalrez costs approximately 5–10× more than Viton per seal, so using it everywhere is not cost-effective. The practical approach is to use Kalrez for process-critical seals (chamber lid, electrode assembly, gas inlet) and Viton for less chemically exposed locations (load-lock, foreline connections, access panel seals).
How do I determine whether particle excursions are caused by chamber contamination or external sources?
Diagnosing particle sources requires a systematic approach. First, run a "static" particle test: place a bare monitor wafer in the chamber without running any plasma, then measure particles. If particles are high, the source is mechanical (wafer handling, load-lock, transfer arm) rather than process-related. Second, run a "dynamic" particle test with the actual process recipe on a bare wafer. If particles appear only after plasma exposure, the source is plasma-related (flaking deposits, electrode erosion, gas-phase nucleation). Third, analyze the spatial distribution of particles: edge-concentrated particles suggest wafer clamping or O-ring issues; center-concentrated particles may indicate showerhead problems; random distribution suggests chamber wall flaking. Fourth, if available, use SEM-EDX (energy-dispersive X-ray spectroscopy) to determine particle composition — aluminum oxide particles point to liner/anodization degradation, silicon particles to electrode erosion, fluorocarbon-rich particles to polymer buildup and flaking, and stainless steel components (Fe, Cr) to mechanical contact or gas delivery issues. This compositional information directly identifies the source component, allowing targeted maintenance rather than a full chamber PM.