Diamond Semiconductor Processing: ICP Etching, Thin Film Deposition & Device Fabrication
By NineScrolls Engineering · 2026-04-01 · 18 min read · Materials Science
Target Readers: Process engineers, device researchers, MEMS/power electronics designers, and procurement teams evaluating equipment for diamond semiconductor fabrication. Engineers developing diamond etching recipes or exploring wide-bandgap device architectures will find the process optimization and equipment selection sections especially relevant.
TL;DR Summary
Diamond is the ultimate wide-bandgap semiconductor — with a 5.47 eV bandgap, the highest thermal conductivity of any material (22 W/cm·K), a breakdown field 30× higher than silicon, and carrier mobilities exceeding most compound semiconductors. These properties make diamond ideal for high-power, high-frequency, and extreme-environment electronics. However, diamond's extreme hardness (10 on the Mohs scale) and chemical inertness demand specialized processing: ICP plasma etching with O₂/Ar chemistry is the leading method for micro/nanostructure fabrication, achieving etch rates of 240–360 nm/min with selectivities up to 1:46 against aluminum masks. This guide covers the fundamental properties that make diamond exceptional, CVD growth methods, ICP etching parameter optimization (gas ratio, ICP power, RF power, chamber pressure), thin-film deposition (DLC via PECVD), doping and contact formation, device architectures, and practical equipment selection guidance.
1. Why Diamond? — The Ultimate Semiconductor Material
Diamond has long been prized for its mechanical and optical properties, but its potential as a semiconductor is now driving a new wave of research and commercialization. Among all known materials, diamond holds simultaneous records for thermal conductivity, hardness, breakdown electric field, and carrier saturation velocity — a combination no other semiconductor can match.
Three converging trends are accelerating diamond semiconductor development:
- CVD growth maturity: High-quality single-crystal diamond (SCD) substrates are now commercially available in sizes up to 10 × 10 mm² at dramatically reduced cost compared to a decade ago, with ongoing efforts toward wafer-scale growth.
- Power electronics demand: SiC and GaN have pushed wide-bandgap adoption, but applications requiring operation above 300 °C, voltages beyond 10 kV, or extreme radiation environments exceed their capabilities — this is where diamond excels.
- Quantum technology: Nitrogen-vacancy (NV) centers in diamond are leading candidates for room-temperature quantum sensors and qubits, creating new demand for precision diamond micro/nanofabrication.
2. Diamond vs. Other Wide-Bandgap Semiconductors
To appreciate diamond's advantages, consider how it compares against established wide-bandgap materials:
| Property | Si | 4H-SiC | GaN | Diamond |
|---|---|---|---|---|
| Bandgap (eV) | 1.12 | 3.26 | 3.39 | 5.47 |
| Breakdown Field (MV/cm) | 0.3 | 3.0 | 3.3 | 10 |
| Thermal Conductivity (W/cm·K) | 1.5 | 4.9 | 1.3 | 22 |
| Electron Mobility (cm²/V·s) | 1,450 | 1,000 | 1,250 | 4,500 |
| Hole Mobility (cm²/V·s) | 480 | 120 | 30 | 3,800 |
| Saturation Velocity (×10⁷ cm/s) | 1.0 | 2.0 | 2.5 | 2.7 |
| Dielectric Constant | 11.7 | 9.7 | 9.0 | 5.7 |
| Max Operating Temp (°C) | 300 | 600 | 400 | >700 |
| BFOM (rel. to Si) | 1 | 340 | 870 | 24,000 |
Table 1: Comparison of key semiconductor properties. BFOM = Baliga's Figure of Merit for power device performance. Diamond's BFOM is ~24,000× that of silicon, indicating its extraordinary potential for high-voltage, low-loss power switching.
Diamond's low dielectric constant (5.7) also benefits high-frequency applications by reducing parasitic capacitance, while its exceptional hole mobility (3,800 cm²/V·s) — the highest of any semiconductor — enables efficient p-type devices, a notable advantage over SiC and GaN where p-type performance is typically poor.
3. CVD Diamond Growth
Modern diamond semiconductor research relies on chemical vapor deposition (CVD) to produce high-purity single-crystal and polycrystalline diamond films. Two primary CVD methods dominate:
3.1 Microwave Plasma CVD (MPCVD)
MPCVD is the gold standard for electronic-grade single-crystal diamond. A hydrogen/methane gas mixture (typically 1–5% CH₄ in H₂) is activated by 2.45 GHz microwave energy, creating a high-density plasma ball above the diamond seed. Atomic hydrogen etches non-diamond carbon phases while methane-derived radicals (CH₃) build the diamond lattice. Growth rates range from 1–30 μm/h depending on conditions, with nitrogen impurity levels below 1 ppb achievable in optimized reactors.
3.2 Hot Filament CVD (HFCVD)
HFCVD uses resistively heated tungsten or tantalum filaments (2,000–2,400 °C) to decompose the gas mixture. It offers lower equipment cost and larger deposition areas than MPCVD, making it suitable for polycrystalline diamond films used in MEMS, thermal management, and optical windows. However, filament contamination (W, Ta incorporation) limits its use for electronic-grade material.
3.3 Substrate Preparation
Typical substrates for diamond device fabrication are 3 × 3 × 0.5 mm³ double-sided polished CVD single-crystal diamonds with initial surface roughness of 5–10 nm. Prior to processing, substrates undergo rigorous chemical cleaning:
- Boiling piranha solution (H₂SO₄:H₂O₂ = 7:3) immersion for 8 hours to remove organic and graphitic contamination
- Sequential ultrasonic cleaning in acetone and anhydrous ethanol
- Nitrogen blow-dry to remove residual solvents
This stringent cleaning protocol ensures a pristine diamond surface free of sp² carbon, metallic contaminants, and adsorbed species that would otherwise compromise subsequent lithographic patterning and plasma etching.
4. ICP Plasma Etching of Diamond
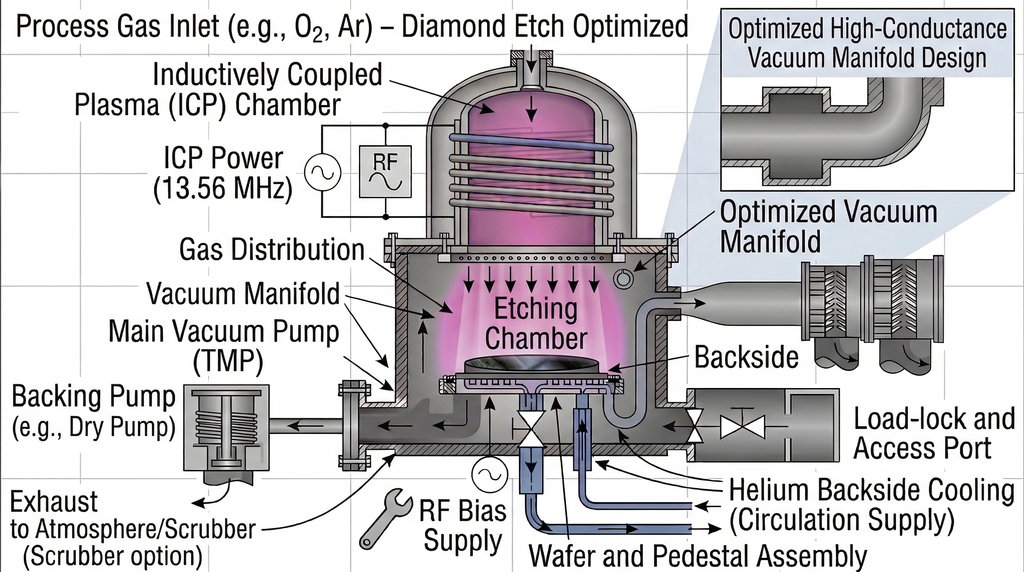
Due to diamond's extreme hardness (Mohs 10) and chemical inertness, conventional mechanical processing cannot achieve the precision required for semiconductor device fabrication. Among the available micro/nanofabrication techniques — laser processing, focused ion beam (FIB), reactive ion etching (RIE), and electron cyclotron resonance (ECR) etching — inductively coupled plasma (ICP) etching has emerged as the preferred method for diamond patterning, offering the best combination of etch rate, precision, surface quality, and process flexibility.
4.1 Why ICP for Diamond?
| Technique | Etch Rate | Precision | Surface Quality | Throughput | Limitations |
|---|---|---|---|---|---|
| Laser Processing | High | Moderate | Poor (cracks, HAZ) | High | Thermal damage, poor dimensional accuracy for micro-features |
| FIB Milling | Very Low | Excellent | Good | Very Low | Ga⁺ implantation, sidewall tilt, prohibitive cost at scale |
| RIE | Low–Moderate | Good | Good | Moderate | Lower plasma density limits etch rate; coupled power/energy control |
| ICP Etching | High | Excellent | Good–Excellent | High | Parameter optimization required; mask selectivity management |
Table 2: Comparison of diamond micro/nanofabrication techniques. ICP etching offers the best overall balance of rate, precision, surface quality, and throughput for semiconductor device patterning.
ICP etching's key advantage is the independent control of plasma density and ion energy. The ICP coil power controls radical/ion generation (plasma density), while the separate RF bias power controls ion bombardment energy at the substrate. This decoupling allows engineers to optimize etch rate and surface quality independently — critical for diamond, where aggressive ion bombardment is needed to break the strong sp³ C–C bonds (bond energy 7.4 eV) but excessive energy degrades surface finish. For a detailed discussion of ICP-RIE architecture and principles, see our ICP-RIE Technology article.
4.2 Etch Chemistry: O₂/Ar
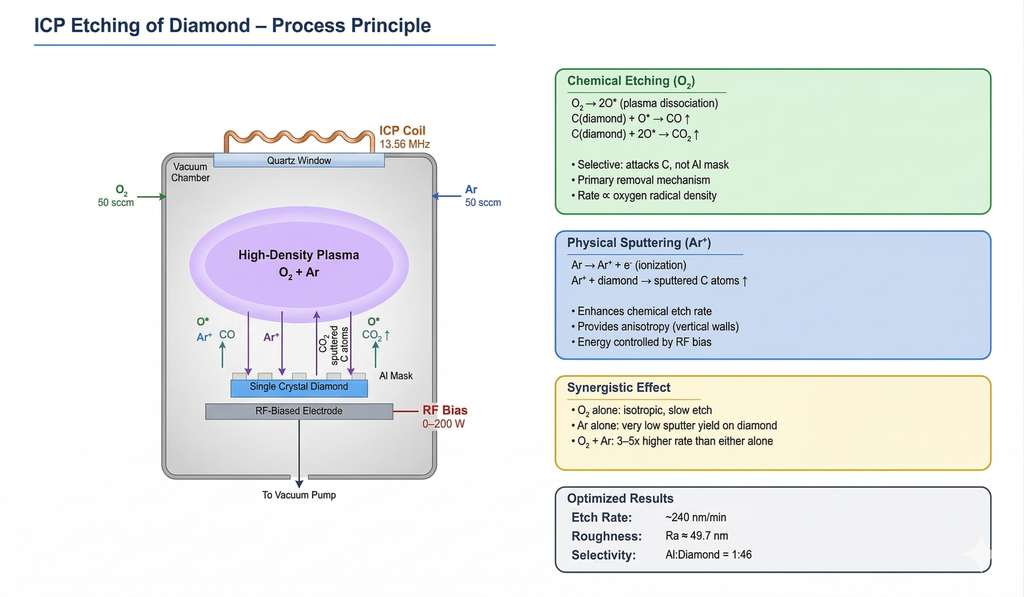
The standard gas chemistry for diamond ICP etching is an O₂/Ar mixture, combining chemical and physical removal mechanisms:
- Chemical pathway (O₂): Reactive oxygen atoms and radicals react with the diamond surface carbon to form volatile CO and CO₂, which are pumped away. This is the primary material removal mechanism and is highly selective — oxygen attacks carbon but not typical mask materials (Al, SiO₂).
- Physical pathway (Ar⁺): Argon ions accelerated by the RF bias physically sputter carbon atoms and oxidized byproducts from the surface, enhancing the chemical etch rate and improving anisotropy (vertical sidewall profile).
The synergy between chemical etching (O₂) and physical bombardment (Ar⁺) is essential: oxygen alone etches isotropically and slowly, while argon alone has extremely low sputter yield against diamond. Together, they achieve etch rates 3–5× higher than either gas alone.
4.3 Mask Strategy
Aluminum deposited via magnetron sputtering is the most common hard mask for diamond ICP etching, offering excellent etch selectivity. The typical process flow:
- Deposit 800–1000 nm aluminum by DC magnetron sputtering
- Spin-coat S1813 positive photoresist, pre-bake
- UV exposure through photomask for pattern transfer
- Develop to reveal aluminum in etch regions
- Wet-etch exposed aluminum (phosphoric/acetic/nitric acid)
- Strip remaining photoresist with acetone and DI water
- ICP etch diamond through patterned aluminum mask
Under optimized conditions, the aluminum-to-diamond etch selectivity reaches 1:46 — meaning 165 nm of aluminum mask supports 7.6 μm of diamond etching. Alternative mask materials include SiO₂, Cr, Ni, and diamond-like carbon (DLC), each offering different selectivity and process compatibility trade-offs.
5. Etching Parameter Optimization
Four key parameters control the ICP diamond etching process. Based on systematic experimental optimization studies[1], the following relationships and optimal values have been established:
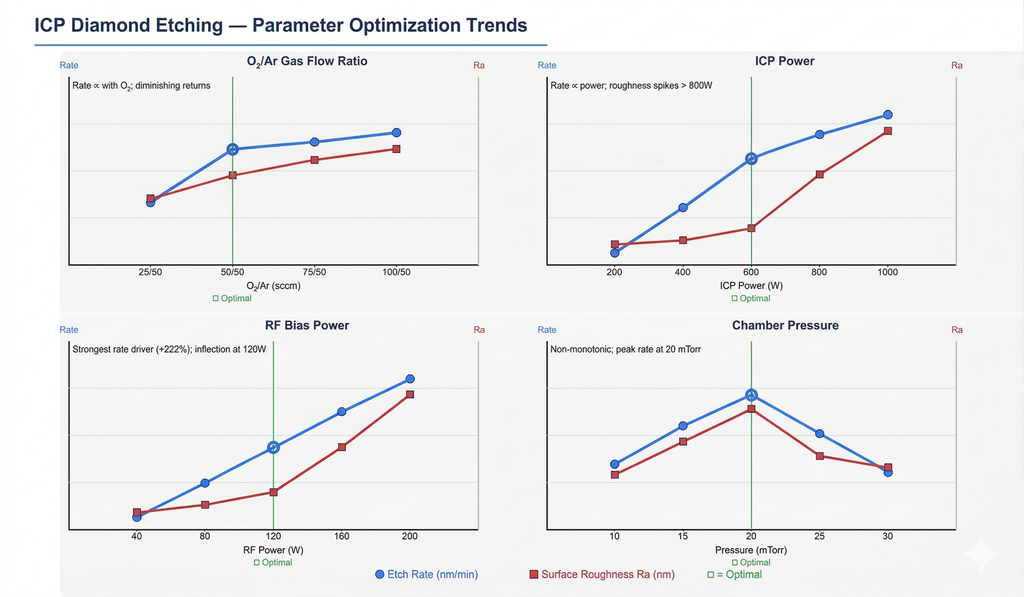
5.1 O₂/Ar Gas Flow Ratio
Effect on etch rate: Increasing the O₂ fraction increases etch rate due to higher reactive oxygen radical concentration. However, excessive total gas flow reduces electron energy and ionization efficiency, creating a diminishing-returns curve.
Effect on surface roughness: Higher O₂ content slightly increases roughness due to crystal-orientation-dependent etch rate variations. Different crystallographic facets of diamond etch at different rates in oxygen plasma, creating nanoscale surface texture.
Optimal value: O₂/Ar = 50/50 sccm — balances a high etch rate (~240 nm/min) with acceptable surface roughness (Ra ≈ 52.5 nm).
5.2 ICP Power
Effect on etch rate: Strong positive correlation. Higher ICP power increases plasma density and reactive radical concentration. Increasing from 200 W to 1000 W raises the etch rate from 94 to 309 nm/min (+230%).
Effect on surface roughness: Stable at low-to-moderate power (200–600 W, Ra ≈ 42–52 nm) but rises sharply above 800 W (Ra > 110 nm) due to intense, non-uniform ion bombardment.
Optimal value: 600 W — delivers a high etch rate while maintaining surface quality in the acceptable range.
5.3 RF Bias Power
Effect on etch rate: Strongest influence of all parameters. Higher RF power increases ion kinetic energy and bombardment intensity. From 40 W to 200 W, the etch rate rises from 113 to 363 nm/min (+222%).
Effect on surface roughness: Gradual increase from 40–120 W, then sharp degradation above 120 W as high-energy ions disrupt the surface structure through excessive sputtering.
Optimal value: 120 W — the inflection point offering the best etch rate vs. roughness trade-off.
5.4 Chamber Pressure
Effect on etch rate: Non-monotonic (increases then decreases). At low pressure, increasing pressure raises plasma density and etch rate. Above 20 mTorr, shortened ion mean free path and reduced ion energy cause the etch rate to decline.
Effect on surface roughness: Relatively small effect overall. Low pressure (10 mTorr) gives the smoothest surface (Ra = 41 nm) due to highly directional ion bombardment.
Optimal value: 20 mTorr — peak etch rate (240 nm/min) with moderate roughness.
5.5 Optimized Parameter Summary
| Parameter | Optimal Value | Range Tested | Key Observation |
|---|---|---|---|
| O₂/Ar Flow | 50/50 sccm | 25–100 / 50 sccm | Higher O₂ = higher rate but diminishing returns; minimal roughness impact |
| ICP Power | 600 W | 200–1000 W | Rate ∝ ICP power; roughness spikes above 800 W |
| RF Bias Power | 120 W | 40–200 W | Strongest rate driver (+222%); 120 W is the roughness inflection point |
| Chamber Pressure | 20 mTorr | 10–30 mTorr | Non-monotonic rate curve; peak at 20 mTorr |
Table 3: Optimized ICP etching parameters for single-crystal diamond patterning (O₂/Ar chemistry, aluminum mask). Data from Zhao et al.[1]
Under these optimized conditions, the process achieves:
- Etch rate: ~240 nm/min
- Surface roughness: Ra ≈ 49.7 nm
- Etch selectivity (Al mask): 1:46
- Etch depth: 7.6 μm in 30 minutes
5.6 Deep Etching Considerations
As etch depth increases, aspect-ratio-dependent effects (ARDE) become significant. In deep diamond trenches, reactive radical transport to the trench bottom becomes diffusion-limited, reducing the local chemical etch rate. Physical argon ion bombardment then becomes the dominant removal mechanism, leading to reduced etch rate, potential profile distortion, and increased surface roughness at depth. For high-aspect-ratio diamond structures (e.g., MEMS resonators, optical waveguides), multi-step recipes with periodic parameter adjustments may be required. For more on ARDE and deep etch strategies, see our DRIE Guide.
6. Diamond-Like Carbon (DLC) Thin Film Deposition
While bulk diamond processing requires etching, many applications benefit from diamond-like carbon (DLC) thin films deposited by plasma-enhanced chemical vapor deposition (PECVD). DLC films combine some of diamond's exceptional properties — high hardness, chemical inertness, low friction, optical transparency — in a thin film that can be deposited on virtually any substrate at moderate temperatures (<300 °C).
6.1 DLC Film Properties
| Property | ta-C (High sp³) | a-C:H (Hydrogenated) | Application Relevance |
|---|---|---|---|
| sp³ Fraction | 80–90% | 40–60% | Higher sp³ → more diamond-like properties |
| Hardness (GPa) | 40–80 | 10–30 | Wear-resistant coatings, hard masks |
| Friction Coefficient | 0.05–0.15 | 0.05–0.20 | MEMS, tribological coatings |
| Optical Bandgap (eV) | 2.0–3.0 | 1.0–2.5 | Optical coatings, IR windows |
6.2 PECVD Deposition of DLC
PECVD systems with dual-frequency RF excitation (13.56 MHz + 400 kHz) provide the best control over DLC film properties. The high-frequency source controls plasma density and dissociation, while the low-frequency source controls ion bombardment energy — directly analogous to the ICP/RF separation in etch systems. Key process parameters:
- Precursor gas: CH₄, C₂H₂, or C₆H₆ diluted in Ar or H₂
- Substrate temperature: 25–300 °C (enables deposition on temperature-sensitive substrates)
- RF power ratio: Higher LF/HF ratio → more ion bombardment → higher sp³ content → harder films
- Pressure: 10–100 mTorr (lower pressure = denser films)
DLC films serve as hard masks for subsequent diamond ICP etching (superior to photoresist for aggressive O₂/Ar chemistries) and as functional coatings for MEMS devices, biomedical implants, and optical components.
Related Equipment: PECVD System — dual-frequency RF design with DLC process capability · ICP Etcher — diamond process design kit available · Contact Us for Diamond Solutions
7. Doping & Ohmic Contact Formation
Realizing diamond semiconductor devices requires controlled doping — introducing impurity atoms to create n-type or p-type conductivity. Diamond's wide bandgap makes this challenging:
7.1 P-type Doping (Boron)
Boron is the only practical p-type dopant for diamond, with an acceptor level 0.37 eV above the valence band. While this is deeper than dopants in Si or GaN, boron-doped diamond still achieves useful conductivity at room temperature, especially at high doping concentrations (>10¹⁹ cm⁻³). At concentrations above ~3 × 10²⁰ cm⁻³, boron-doped diamond becomes metallic (superconducting below ~4 K) — useful for electrode applications.
Doping methods: In-situ during CVD growth (B₂H₆ or TMB added to the gas mixture) is preferred over ion implantation, as implantation severely damages the diamond lattice and requires high-temperature annealing (>1400 °C) to recover crystallinity.
7.2 N-type Doping (Phosphorus, Nitrogen)
N-type doping remains diamond's greatest challenge. Nitrogen — abundant and easily incorporated — has a donor level 1.7 eV below the conduction band, making it electrically inactive at room temperature. Phosphorus (donor level 0.57 eV) is the best available n-type dopant, but its incorporation efficiency into the diamond lattice is low and achievable concentrations are limited. This asymmetry between p-type and n-type doping has steered most diamond device research toward unipolar (p-type) architectures.
7.3 Ohmic Contact Formation
Low-resistance ohmic contacts to diamond are achieved using carbide-forming metals:
- P-type contacts: Ti/Pt/Au or Ti/Au stacks annealed at 400–500 °C. Titanium reacts with the diamond surface to form TiC, creating a low-barrier interface.
- N-type contacts: Ti/Pt/Au on phosphorus-doped surfaces, though contact resistances remain higher than for p-type due to lower doping levels.
- Hydrogen-terminated surfaces: Oxygen-terminated diamond surfaces are insulating, while hydrogen-terminated surfaces exhibit a 2D hole gas (2DHG) with sheet carrier densities of ~10¹³ cm⁻², enabling surface-channel FETs without bulk doping.
8. Diamond Device Architectures & Applications
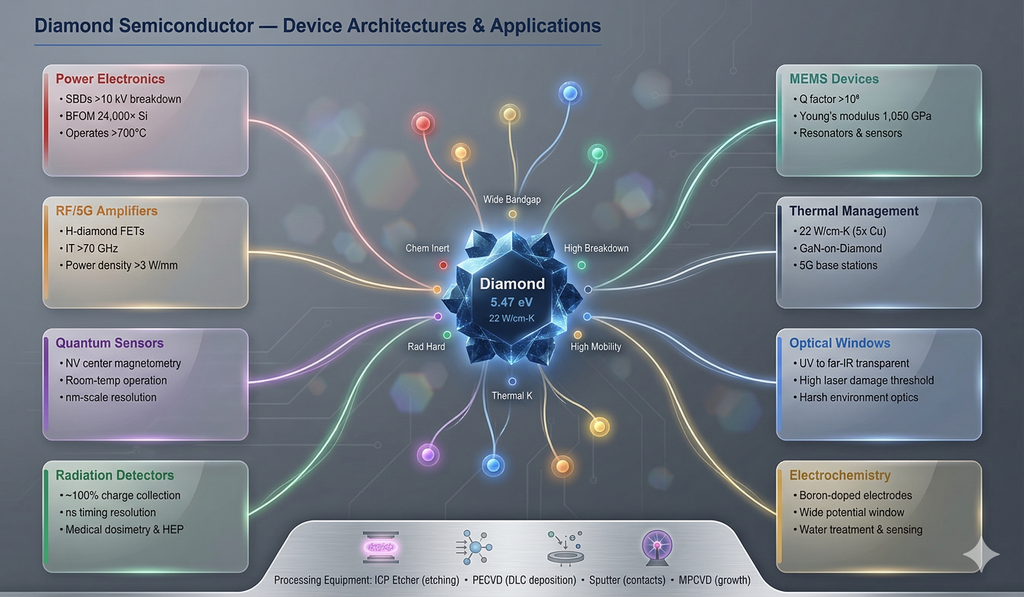
8.1 Power Schottky Barrier Diodes (SBDs)
Diamond Schottky diodes using boron-doped drift layers and high-work-function metals (Pt, Mo, WC) have demonstrated breakdown voltages exceeding 10 kV and on-resistances below those predicted by the SiC unipolar limit. These devices operate at temperatures above 400 °C, making them candidates for geothermal, automotive, and aerospace power conversion where silicon and even SiC devices cannot survive.
8.2 Field-Effect Transistors (FETs)
Two main FET architectures exploit diamond's properties:
- Hydrogen-terminated diamond FETs (H-diamond FETs): Utilize the surface 2D hole gas created by hydrogen termination and surface adsorbates. These devices have achieved RF power densities >3 W/mm and cutoff frequencies >70 GHz, positioning diamond for 5G/6G and satellite communication amplifiers.
- Deep-depletion MOSFETs: Use high-κ gate dielectrics (Al₂O₃, ZrO₂) on boron-doped diamond channels. Gate oxide quality and interface trap density remain active research areas.
8.3 Radiation Detectors
Diamond's radiation hardness, low atomic number (Z = 6), and tissue-equivalence make it an ideal material for dosimetry in medical radiation therapy and high-energy physics. Single-crystal CVD diamond detectors exhibit near-100% charge collection efficiency, nanosecond timing resolution, and stability after exposure to >10¹⁵ protons/cm². ICP-etched 3D electrode structures in diamond improve detector response uniformity and radiation tolerance for LHC-class experiments.
8.4 Quantum Sensors (NV Centers)
Nitrogen-vacancy (NV) centers in diamond are atomic-scale quantum sensors for magnetic field, electric field, temperature, and strain — operating at room temperature, unlike most quantum systems. Precision ICP etching is used to fabricate diamond nanopillars, photonic crystal cavities, and cantilevers that enhance NV center optical collection efficiency by 10–20× and enable scanning probe magnetometry with nanometer resolution.
8.5 MEMS & Microfluidics
Diamond's combination of high Young's modulus (1,050 GPa), low internal friction, and chemical inertness makes it exceptional for MEMS resonators (Q factors >10⁶ at GHz frequencies), pressure sensors for corrosive environments, and microfluidic channels for harsh chemical processing. ICP etching enables fabrication of these complex 3D microstructures with sub-micron precision.
8.6 Thermal Management
Diamond's unmatched thermal conductivity (22 W/cm·K — 5× copper, 15× silicon) makes it the ultimate heat spreader for high-power-density electronics. Diamond heat spreaders bonded to GaN HEMTs reduce channel temperatures by 40–60 °C, enabling higher output power and improved reliability. Polycrystalline diamond grown directly on GaN-on-SiC wafers (GaN-on-diamond technology) is entering commercial production for 5G base station amplifiers.
9. Equipment Selection Guide
Diamond processing places unique demands on fabrication equipment. Here is a practical guide to selecting the right tools:
9.1 ICP Etcher Requirements for Diamond
- ICP source power: ≥600 W minimum; 1000+ W preferred for production throughput. Higher power enables the high radical density needed for reasonable diamond etch rates.
- RF bias power: Independently controllable, 0–200+ W range. The 120 W optimum for diamond requires precise, low-noise bias control.
- Gas delivery: O₂ and Ar with individual mass flow controllers (MFCs). Additional lines for CF₄, Cl₂, or BCl₃ enable processing of other materials in the same chamber.
- Temperature control: Helium backside cooling with temperature range −20 °C to 200 °C. Diamond etch uniformity is sensitive to substrate temperature.
- Chamber materials: Alumina or anodized aluminum chamber liners resistant to oxygen plasma.
- Endpoint detection: Laser interferometry and/or optical emission spectroscopy (OES) for precise etch depth control.
- Process library: A diamond etch process design kit (PDK) accelerates recipe development and reduces qualification time.
9.2 PECVD Requirements for DLC
- Dual-frequency RF: 13.56 MHz + 400 kHz for independent control of plasma chemistry and ion energy — essential for tuning DLC sp³ fraction.
- Hydrocarbon gas capability: CH₄ or C₂H₂ lines with appropriate safety interlocks.
- Low-temperature operation: Substrate temperature control down to room temperature for deposition on temperature-sensitive devices.
- Film stress control: Tunable film stress (compressive to tensile) via power ratio and pressure adjustment.
NineScrolls Diamond Processing Solutions: ICP Etcher Series — up to 8 gas lines, diamond PDK, He backside cooling, laser interferometry endpoint · PECVD System — dual-frequency RF, DLC-capable, room-temperature to 350 °C · Request a Diamond Processing Consultation
10. Frequently Asked Questions
Q: What etch rate can I expect for single-crystal diamond using ICP?
A: With optimized O₂/Ar chemistry (50/50 sccm, ICP 600 W, RF 120 W, 20 mTorr), etch rates of ~240 nm/min are achievable with Ra ≈ 50 nm roughness. Increasing RF power to 200 W can push rates above 360 nm/min, but at the cost of significantly higher surface roughness. The optimal trade-off depends on your application's roughness tolerance.
Q: Why is ICP preferred over RIE for diamond etching?
A: ICP provides 2–5× higher plasma density than capacitively coupled RIE at the same pressure, which directly translates to higher etch rates for chemically-resistant materials like diamond. Additionally, the independent control of ICP power (plasma density) and RF bias (ion energy) allows optimization of etch rate and surface quality separately — a degree of freedom that coupled CCP-RIE systems lack.
Q: Can polycrystalline diamond be etched with the same process?
A: Yes, but with caveats. Polycrystalline diamond contains grain boundaries and mixed crystal orientations, leading to orientation-dependent etch rates and higher post-etch surface roughness compared to single-crystal material. The same O₂/Ar chemistry and parameter ranges apply, but recipe optimization for polycrystalline diamond focuses more heavily on minimizing grain-boundary-related roughness.
Q: What is the best mask material for deep diamond etching?
A: Aluminum is the most practical choice, offering ~1:46 selectivity. For deeper etches (>20 μm), consider thicker Al masks (2+ μm), SiO₂ hard masks deposited by PECVD, or electroplated nickel which can achieve selectivities >1:100 in some chemistries. The choice depends on pattern resolution requirements and downstream process compatibility.
Q: Is diamond ready for commercial semiconductor production?
A: Diamond is currently in the transition from R&D to early commercialization. Commercial products exist in niche applications (radiation detectors, high-power RF amplifiers, thermal management substrates), while broader adoption awaits solutions to substrate size limitations and n-type doping challenges. The GaN-on-diamond thermal management platform is the nearest-term high-volume application.
Q: What surface roughness is acceptable for diamond electronic devices?
A: For RF FETs and Schottky diodes, Ra < 1 nm (CMP-finished) is typically required on the active device surface. ICP etch roughness of ~50 nm is acceptable for MEMS structures, waveguide trenches, and mesa isolation features. Critical device surfaces may require post-etch CMP polishing.
References
- Zhao, L., Wang, X., Jiang, N. et al. "Experimental study of inductively coupled plasma etching of patterned single crystal diamonds." Scientific Reports 15, 21062 (2025). doi:10.1038/s41598-025-08066-3
- Donato, N., Rouger, N., Pernot, J., Longobardi, G. & Udrea, F. "Diamond power devices: state of the art, modelling, figures of merit and future perspective." J. Phys. D 53, 093001 (2019).
- Isberg, J., Hammersberg, J., Twitchen, D. J. & Whitehead, A. J. "Single crystal diamond for electronic applications." Diam. Relat. Mater. 13, 320–324 (2004).
- Luo, J. K. et al. "Diamond and diamond-like carbon MEMS." J. Micromech. Microeng. 17, S147–S163 (2007).
- Liu, J. et al. "Low on-resistance diamond field effect transistor with high-k ZrO₂ as dielectric." Sci. Rep. 4, 6395 (2014).
- Lu, J., Xu, D., Huang, N., Jiang, X. & Yang, B. "One-dimensional diamond nanostructures: fabrication, properties and applications." Carbon 223, 119020 (2024).
- Tran, D. T., Fansler, C., Grotjohn, T. A., Reinhard, D. K. & Asmussen, J. "Investigation of mask selectivities and diamond etching using microwave plasma-assisted etching." Diam. Relat. Mater. 19, 778–782 (2010).